Key takeaways
Semaglutide is a type of prescription medication called a GLP-1 receptor agonist.
Semaglutide works by mimicking a naturally occurring hormone that regulates blood sugar, reduces appetite, and increases satiety (how full and satisfied you feel from food).
Semaglutide may be prescribed for type 2 diabetes, or to promote weight loss in people with obesity or overweight.
It is available in three brand names and formulas: Ozempic, Wegovy, and Rybelsus.
Here's what we'll cover
Here's what we'll cover
Here's what we'll cover
Key takeaways
Semaglutide is a type of prescription medication called a GLP-1 receptor agonist.
Semaglutide works by mimicking a naturally occurring hormone that regulates blood sugar, reduces appetite, and increases satiety (how full and satisfied you feel from food).
Semaglutide may be prescribed for type 2 diabetes, or to promote weight loss in people with obesity or overweight.
It is available in three brand names and formulas: Ozempic, Wegovy, and Rybelsus.
Semaglutide is the active ingredient in three brand-name prescription drugs–Ozempic, Wegovy, and Rybelsus–all of which are glucagon-like peptide 1 receptor agonists, commonly called GLP-1s. This class of drugs has become extremely popular due to their effectiveness in both regulating blood sugar and promoting weight loss.
If you’re curious about using semaglutide, you might have questions about its potential benefits and risks, and whether it’s right for you. We can help.
Read on to learn how semaglutide works, what it’s used for, its effectiveness for blood sugar regulation and weight loss, safety considerations, and more.
How does semaglutide work?
GLP-1 medications like semaglutide work by mimicking the action of the GLP-1 hormone, a type of incretin hormone that is produced in the gut and helps stimulate insulin secretion after you eat. Insulin is a hormone that is made in the pancreas. When you eat, insulin converts glucose (sugar) into energy, which helps to regulate blood sugar levels.
Semaglutide also binds to several GLP-1 receptors in the body that may promote weight loss and reduce risks for cardiovascular disease. Here’s a breakdown of how semaglutide works in different parts of the body:
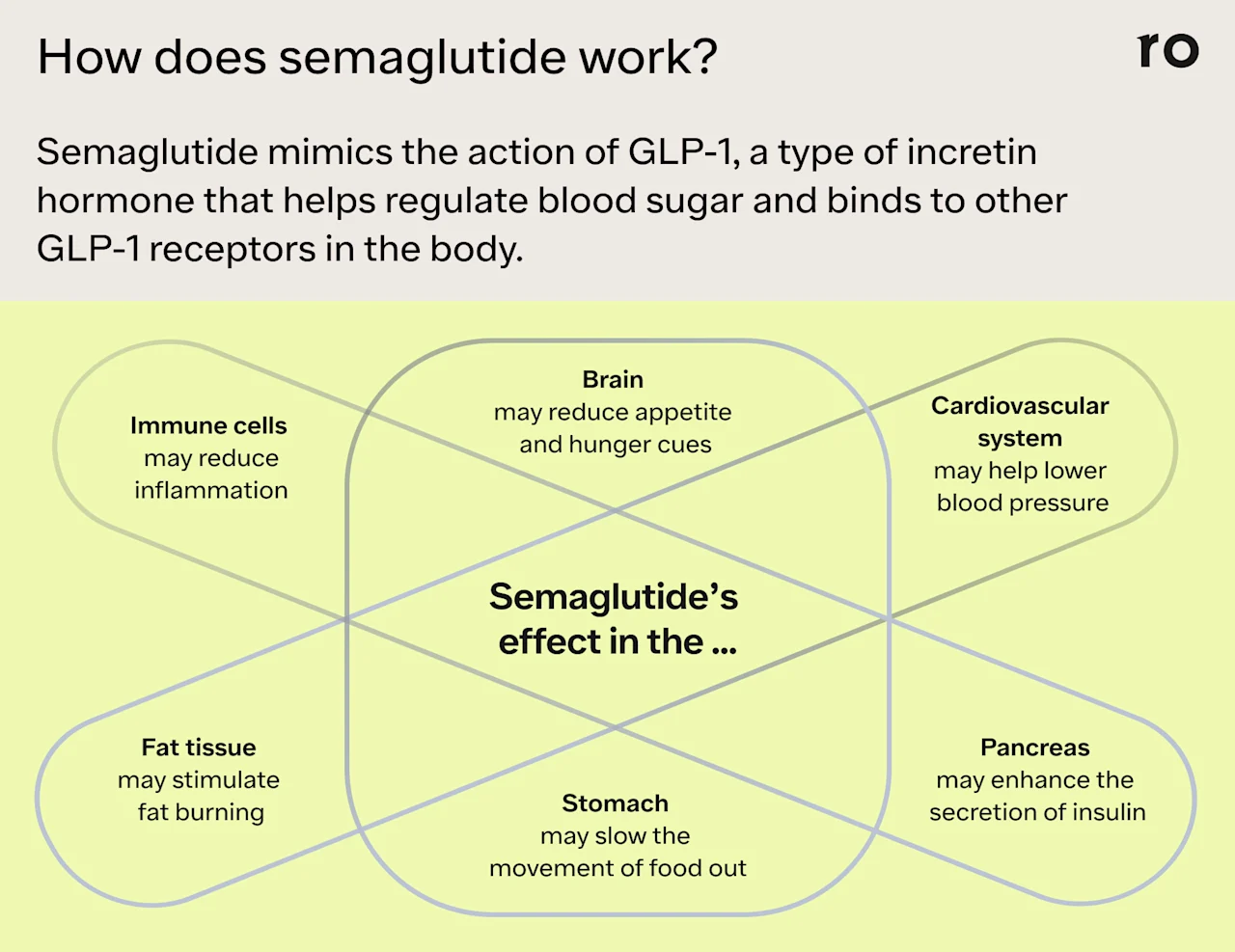
Brain
Semaglutide impacts appetite centers in the brain to reduce hunger cues and lessen appetite. Many people also report less “food noise,” which means they have to deal with fewer intrusive thoughts surrounding food, helping them eat less.
Pancreas
GLP-1s stimulate the secretion of insulin, a hormone that helps move sugar(glucose) from your bloodstream and into your cells, where it can be turned into energy. This lowers blood glucose levels. Semaglutide also suppresses glucagon secretion. Glucagon is a hormone produced in the pancreas that sends messages to the liver to release stored sugar and raise blood sugar levels.
Stomach
Semaglutide slows gastric emptying, or the rate at which food moves out of the stomach and into the small intestine. It also promotes the release of satiety hormones. This helps you feel fuller after eating and more satisfied with meals, which reduces the amount of food you eat.
Cardiovascular system
Semaglutide and other GLP-1 receptor agonists have been shown to help lower blood pressure and reduce overall cholesterol levels. GLP-1 agonists may also help the heart pump better, improving blood flow and decreasing the risk of heart-related health problems.
Immune cells
Some research suggests that semaglutide may help reduce oxidative stress (which can cause cell damage)and inflammation, both of which may contribute to obesity.
Fat tissue
Semaglutide also enhances energy expenditure. This can stimulate fat burning and discourage fat storage, which may aid in weight loss.
How does semaglutide help you lose weight?
Semaglutide promotes weight loss by:
Regulating blood sugar, helping your body convert glucose into energy
Decreasing hunger cues and “food noise” in the brain, leading to a lower appetite
Delaying stomach emptying, helping you to feel full faster and more satisfied after meals
Increasing energy expenditure and fat burning, which can help reduce body fat and lipids
Types of semaglutide: comparing Ozempic, Wegovy, and Rybelsus
Semaglutide is available under three brand names: Ozempic, Wegovy, and Rybelsus.
The FDA initially approved semaglutide under the brand name Ozempic in 2017 for managing type 2 diabetes, though it is sometimes prescribed off-label for weight loss. Off-label means that a drug is prescribed for a purpose other than its intended indication.
Semaglutide was later approved under the brand names Rybelsus (type 2 diabetes) and Wegovy (weight loss).
Here’s a quick breakdown of each brand, its indications, and dosing information.

Ozempic
Ozempic is FDA-approved and indicated for:
Improving blood sugar control in adults with type 2 diabetes, alongside diet and exercise
Reducing the risk of major cardiovascular events (like heart attack and stroke) in adults with type 2 diabetes who also have cardiovascular disease
It may also be prescribed off-label for weight loss.
Ozempic is a self-injectable medication that you take once a week, on the same day each week. It comes in a pre-filled pen that you inject into your abdomen, the front of your thigh, or the back of your upper arm.
The starting dose is 0.25 milligrams. After about four weeks, your healthcare provider will slowly increase your dose and continue to do so each month until you reach the desired level of blood sugar control. Your provider will monitor any side effects or other health impacts during this time and may adjust your dosage depending on how well you tolerate Ozempic.
Your healthcare provider can guide you on the goal range for your blood sugar levels and A1C. The maintenance (ongoing) dosage of Ozempic is 0.5 mg, 1 mg, or 2 mg once weekly. The drug is usually prescribed for long-term treatment, assuming it continues to be safe and effective for you.
Ozempic dosing schedule:
Weeks 1-4: Starting dose 0.25 mg once weekly
Weeks 5-8: 0.5 mg once weekly
Weeks 9-12: 1 mg once weekly
Weeks 13-16: 2 mg once weekly
Wegovy
Wegovy is FDA-approved for weight management alongside diet and exercise in people with obesity, or people who have overweight and other weight-related health conditions. Wegovy is indicated for people ages 12 years and older who have:
A BMI of 30 kg/m2 or higher
A BMI of 27 kg/m2 to 29.9 kg/m2 and at least one weight-related comorbidity, such as high blood pressure, type 2 diabetes, or obstructive sleep apnea
Wegovy is also indicated to reduce the risk of major cardiovascular events, such as heart attack or stroke, in people who have obesity or overweight.
Wegovy is a self-injectable medication that you take once a week, on the same day each week. It comes in a single-use pen that you inject into your abdomen, thigh, or upper arm. It comes in five dose strengths: 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg.
Like Ozempic, dosing starts at the lowest possible dose and gradually increases over time and under your healthcare provider’s guidance. The long-term maintenance dosage of Wegovy is either 1.7 mg or 2.4 mg once weekly, depending on individual factors.
Wegovy dosing schedule:
Weeks 1-4: Starting dose 0.25 mg once weekly
Weeks 5-8: 0.5 mg once weekly
Weeks 9-12: 1 mg once weekly
Weeks 13-16: 1.7 mg once weekly
Weeks 17+: 1.7 or 2.4 mg once weekly
The recommended maintenance dose of Wegovy in adults is 1.7 or 2.4 mg once weekly. For pediatric patients ages 12 and over, the maintenance dose is 2.4 mg once weekly.
Rybelsus
Rybelsus is FDA-approved for controlling blood sugar levels in adults with type 2 diabetes in addition to lifestyle changes like diet and exercise.
Unlike other semaglutide-based medications, Rybelsus comes in an oral tablet, not an injectable solution.
Rybelsus is a once-daily pill that is available in three dose strengths: 3 mg, 7 mg, and 14 mg. You’ll typically start at 3 mg once a day for 30 days before increasing your dose to 7 mg. Depending on how well you tolerate the medication, any side effects you experience, and your blood sugar control, your healthcare provider may increase the dosage of Rybelsus to the maximum dose of 14 mg once daily.
The timing of Rybelsus matters. You should take Rybelsus on an empty stomach and with plain water only, but no more than 4 ounces. Then you should wait at least 30 minutes before your first food, drink, or other oral medications of the day. Not following these instructions can make Rybelsus less effective or more likely to cause side effects.
Rybelsus dosing schedule:
First 30 days: Starting dose 3 mg once daily
Days 31+: Increase to 7 mg once daily
Days 61+: Increase to 14 mg once daily, if needed
How much weight can you lose with semaglutide?
The amount of weight a person can lose with semaglutide and other GLP-1s will depend on factors like dosing, duration of use, and whether or not it's paired with lifestyle interventions like diet and exercise.
That said, some clinical trials on semaglutide provide a glimpse at potential weight loss results.
These are key statistics from one double-blind trial on 1,961 adults who received a once-weekly injection of semaglutide at a dose of 2.4 mg for 68 weeks. Participants in the treatment group paired the medication with a reduced-calorie diet (eating around 500 fewer calories per day) and 150 minutes of physical activity each week.
On average, people taking semaglutide lost 14.9% of their body weight, compared to a 2.4% body weight loss in the placebo group.
Those on semaglutide lost an average of 33.7 pounds compared to 5.7 pounds in the placebo group.
The majority of participants in the treatment group (86.4%) lost 5% of their body weight or more compared to 31.5% on placebo. Even this lower percentage of weight loss may be associated with metabolic health benefits, lower blood pressure, and reduced lipids.
50.5% lost 15% or more of their body weight compared to 4.9% of the placebo group.
Another trial on 1,210 people found similarly promising results, with those taking 2.4 mg of semaglutide reducing their body weight by 9.6% compared to 3.4% in the placebo group over 68 weeks of treatment.
One longer-term study followed people taking semaglutide for 208 weeks (about 4 years). In that treatment group, the average weight loss was 10.2% on a once-weekly dose of 2.4 mg. Participants also lost about three inches off of their waist circumference compared to the placebo group.
There are also potential heart-health benefits to taking semaglutide. A 2023 study found a 20% cardiovascular risk reduction for obese individuals taking semaglutide compared to those not taking a GLP-1 drug.
Sustainable weight loss with semaglutide
While significant weight loss is possible with semaglutide, it’s important to remember that these medications are not “miracle” drugs. They are designed to be used long-term and in combination with intensive lifestyle modifications–including a reduced-calorie diet and 150 minutes of exercise per week– to achieve and maintain significant and sustainable weight loss.
What are the side effects of semaglutide?
Although the health benefits of these medications may be impressive, they may not be appropriate for everyone. Semaglutide comes with side effects or possible adverse health effects, depending on your family and personal health history.
“These medications are appropriate when their benefits outweigh their risks,” says Charlie Seltzer, MD, a board-certified obesity medicine doctor in Philadelphia. He adds that it’s important to talk to your provider about your health goals, personal and family health history, and any medications you might be taking. This information can help your HCP determine if semaglutide is a good option for you.
The most common side effects of semaglutide include:
Nausea
Vomiting
Diarrhea
Constipation
Abdominal pain
Pain or itching at the injection site (with Ozempic or Wegovy)
The GI-related side effects occur more frequently when you first start on semaglutide or when you increase your dosage. In most cases, nausea and other digestive side effects go away as your body adjusts to the medication.
Tell your healthcare provider if your side effects seem severe, or if they don’t go away over time. Your provider may reduce your dose, suggest a longer timeline before increasing your dose, or recommend other ways to manage these side effects.
Some ways to manage common side effects might include:
Starting at the lowest possible dose and gradually increasing it over time
Taking prescription or over-the-counter anti-nausea medications
Eating smaller, more frequent meals
Drinking plenty of water
Warnings and precautions
Serious adverse effects are rare but have been reported with semaglutide. If you experience symptoms of the following adverse effects, stop taking semaglutide and contact your healthcare provider:
Thyroid tumors: Semaglutide may increase the risk for certain types of thyroid tumors including medullary thyroid cancer or multiple endocrine neoplasia syndrome 2 (MEN2). Call your provider if you feel a lump in your throat, have a hoarse voice, or have trouble swallowing.
Pancreatitis (inflammation of the pancreas): Symptoms may include severe upper abdominal pain that doesn’t go away, vomiting, and nausea
Gallbladder disease (gallstones or gallbladder inflammation): Symptoms may include sudden and intense abdominal pain, nausea, vomiting, fever and chills
Kidney problems: Changes in urination (either going more or less frequently), along with swelling in feet and ankles
Hypersensitivity or allergic reactions (including anaphylaxis): Shortness of breath, itching and swelling near the injection site, dizziness
Diabetic retinopathy: People with type 2 diabetes and a history of diabetic retinopathy (a complication that affects the blood vessels in the eye) may experience worsening symptoms, including blurred vision, spots, and floaters
Low blood sugar (hypoglycemia): Symptoms include fast heartbeat, shakiness, dizziness, and confusion. Semaglutide may cause hypoglycemia when taken with certain diabetes medications, such as insulin or sulfonylureas
This is not a complete list of all warnings and precautions for semaglutide. Talk to your provider if you have any concerns or questions about a medication you’re taking.
Who should not take semaglutide?
Due to potential health risks and complications, some people should not take semaglutide. That includes:
People who are pregnant or planning to become pregnant
People with a personal or family history of kidney problems
People with a personal or family history of pancreatitis
People with a personal or family history of medullary thyroid cancer
People who have a known allergy or hypersensitivity to the ingredients in semaglutide
People who are already taking a GLP-1 weight loss medication
Always talk to your provider about potential risks and health complications before starting a new medication. Based on factors like your personal and family health history, they may determine that semaglutide is not a good option for you.
How does semaglutide compare to tirzepatide?
Tirzepatide is a dual-agonist GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) medication that may be prescribed for blood sugar regulation and weight loss. It is available under the brand names Mounjaro (type 2 diabetes) and Zepbound (weight loss). These medications also come in self-injectable pens and varying dose strengths.
Side effects for tirzepatide are also similar to those for semaglutide and include mild and manageable GI issues like nausea, vomiting, diarrhea, and constipation.
The mechanism of action for tirzepatide is similar to semaglutide, but because tirzepatide also activates GIP receptors it may be more effective for blood sugar control and weight loss. When comparing those two medications head-to-head, tirzepatide resulted in a 6.9% greater body weight reduction versus semaglutide over 12 months.
Like semaglutide, tirzepatide is also designed to be used long-term and combined with diet and exercise to achieve sustainable results.
Remember that your results with either medication will vary depending on personal health, lifestyle factors, and medication dosing and duration. Talk to your provider about any questions you might have, and which medication might be right for you.
Long-term use of semaglutide for weight loss
Semaglutide, like other GLP-1 medications, is designed to be taken long-term. The majority of clinical trials followed people taking semaglutide for one to two years. The exact amount of time that you will take semaglutide depends on several factors, including your weight loss goals, your overall health status, and your provider’s recommendation.
“There is a common narrative on social media right now that GLP medications can be used to ‘jumpstart’ your weight loss goals. While that may be the case for some people, most people will find that using the medications long-term is more supportive of their goals,” says Sarah Stombaugh, MD, a family medicine physician and diplomate of the American Board of Obesity Medicine.
What’s more, stopping semaglutide too soon may lead to rebound weight gain or high blood sugar. If you’re considering taking semaglutide, keep in mind that you will need to take it for a long time to maintain weight loss and blood sugar control.
Dr. Stombaugh adds that some people may be able to decrease their dose or spread out the time between injections, but those decisions are made on a case-by-case basis and in close collaboration with your healthcare provider. They can help you determine the optimal dosing and timing of these weight loss injections.
Bottom line
Semaglutide is a GLP-1 medication available in three formulas: Ozempic, an injectable medication to treat diabetes; Wegovy, an injectable medication for weight loss; and Rybelsus, an oral diabetes medication.
GLP-1 medications mimic a naturally occurring hormone that reduces appetite, delays gastric emptying, and lowers blood sugar.
Clinical trials show that taking a 2.4 mg once-weekly dose of semaglutide can lead to about a 10 to 15% reduction in overall body weight over 68 weeks. Studies show that with long-term use, people can maintain this weight loss for years, though you need to continue to take the medication.
Compared to other medications, semaglutide has been found to be more effective for weight loss than liraglutide (Saxenda), though not as effective as tirzepatide (Zepbound). Semaglutide may lead to greater weight loss than some oral weight loss medications.
To learn more about whether semaglutide is the right option for you to treat diabetes or aid in weight loss alongside diet and exercise, talk to a provider who is knowledgeable in weight loss treatments.
Ro has helped over 80,000 members lose weight with GLP-1s. Connect with one of our medical experts today to learn more.
Frequently asked questions (FAQs)
How does semaglutide work for weight loss?
Semaglutide works on several fronts:
In appetite centers of the brain to decrease hunger cues and lower appetite
In the stomach to slow gastric emptying and help you feel full faster and longer
In the pancreas, stimulating insulin and suppressing glucagon to lower blood sugar levels
What does semaglutide do?
Semaglutide mimics the GLP-1 hormone in the body. This hormone serves a variety offunctions, including:
Triggering insulin production in the pancreas which enables glucose to leave the bloodstream and move into the cells. Glucose (sugar) in the cells can be used for energy. This process helps regulate blood sugar levels
Suppressing glucagon secretion, which also helps to lower blood sugar
Slowing gastric emptying, making food move more slowly out of your stomach and helping you feel full faster and for longer periods
Quieting hunger cues in the brain to reduce appetite
How long does it take for semaglutide to kick in?
Weight loss results from semaglutide are individual and depend on factors like dosage and lifestyle habits.
According to the manufacturer, there is a steady level of semaglutide in the bloodstream within four to five weeks of once-weekly injections; however, it’s unclear if this means you’ll start seeing weight loss results in the same amount of time.
That said, some research shows that people on semaglutide can lose up to 5.9% of their body weight after three months of treatment.
How does semaglutide reduce belly fat?
A reduction in belly fat likely comes from a weight reduction overall. Semaglutide may also directly aid in fat burning and boost energy expenditure, which can impact weight loss.
How much does semaglutide cost?
The cost of semaglutide varies depending on which brand you are taking, and factors like insurance coverage, savings programs, and coupons. The current list prices (without insurance or savings programs) for Ozempic, Wegovy, and Rybelsus are:
Ozempic: $968.52
Wegovy: $1349.02
Rybelsus: $968.52
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
Ozempic Important Safety Information: Read more about serious warnings and safety info.
Wegovy Important Safety Information: Read more about serious warnings and safety info.
GLP-1 Important Safety Information: Read more about serious warnings and safety info.
Mounjaro Important Safety Information: Read more about serious warnings and safety info.
Zepbound Important Safety Information: Read more about serious warnings and safety info.
Saxenda Important Safety Information: Read more about serious warnings and safety info.
References
Collins L. & Costello R.A. (2024). Glucagon-Like Peptide-1 Receptor Agonists. StatPearls. Retrieved Nov. 1, 2024 from https://www.ncbi.nlm.nih.gov/books/NBK551568/
Davies, M., Færch, L., Jeppesen, O. K., et al. (2021). Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet (London, England), 397(10278), 971–984. doi: 10.1016/S0140-6736(21)00213-0. Retrieved from https://pubmed.ncbi.nlm.nih.gov/33667417/
Filippatos, T. D., Panagiotopoulou, T. V., & Elisaf, M. S. (2014). Adverse Effects of GLP-1 Receptor Agonists. The Review of Diabetic Studies : RDS, 11(3-4), 202–230. doi: 10.1900/RDS.2014.11.202. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC5397288/
Ghusn, W., De la Rosa, A., Sacoto, D., et al. (2022). Weight Loss Outcomes Associated With Semaglutide Treatment for Patients With Overweight or Obesity. JAMA Network Open, 5(9), e2231982. doi: 10.1001/jamanetworkopen.2022.31982. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC9486455
Lincoff, A. M., Brown-Frandsen, K., Colhoun, H. M., et al. (2023). Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. The New England Journal of Medicine, 389(24), 2221–2232. doi: 10.1056/NEJMoa2307563. Retrieved from https://pubmed.ncbi.nlm.nih.gov/37952131/
Novo Medlink. (2024). Naviating Coverage Eligibility. Retrieved from https://www.novomedlink.com/obesity/products/treatments/wegovy/cost-coverage/wegovy-coverage-savings.html
Novo Medlink. (2023). Once-weekly Ozempic (semaglutide) injection mechanism of action. Retrieved from https://www.novomedlink.com/diabetes/products/treatments/ozempic/about/mechanism-of-action.html
Novo Nordisk. (2023). Ozempic Highlights of Prescribing Information. Novo Nordisk. Retrieved from https://www.novo-pi.com/ozempic.pdf
Novo Nordisk-a. (2024). Rybelsus Highlights of Prescribing Information. Novo Nordisk. Retrieved from https://www.novo-pi.com/rybelsus.pdf
Novo Nordisk-b. (2024). Wegovy Highlights of Prescribing Information. Novo Nordisk. Retrieved from https://www.novo-pi.com/wegovy.pdf
Rodriguez, P. J., Goodwin Cartwright, B. M., Gratzl, S., et al. (2024). Semaglutide vs Tirzepatide for Weight Loss in Adults With Overweight or Obesity. JAMA Internal Medicine, 184(9), 1056–1064. doi: 10.1001/jamainternmed.2024.2525. Retrieved from https://pubmed.ncbi.nlm.nih.gov/38976257/
Ryan, D. H., Lingvay, I., Deanfield, J., et al. (2024). Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nature Medicine, 30(7), 2049–2057. doi: 10.1038/s41591-024-02996-7. Retrieved from https://pubmed.ncbi.nlm.nih.gov/38740993/
Seufert, J. & Gallwitz, B. (2014). The extra-pancreatic effects of GLP-1 receptor agonists: a focus on the cardiovascular, gastrointestinal and central nervous systems. Diabetes, Obesity & Metabolism, 16(8), 673–688. doi: 10.1111/dom.12251. Retrieved from https://pubmed.ncbi.nlm.nih.gov/24373150/
Tamayo-Trujillo, R., Ruiz-Pozo, V. A., Cadena-Ullauri, S., et al. (2024). Molecular mechanisms of semaglutide and liraglutide as a therapeutic option for obesity. Frontiers in Nutrition, 11, 1398059. doi: 10.3389/fnut.2024.1398059. Retrieved from https://pubmed.ncbi.nlm.nih.gov/38742021/
Wilding, J. P. H., Batterham, R. L., Calanna, S.,et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity. The New England Journal of Medicine, 384(11), 989–1002. doi: 10.1056/NEJMoa2032183. Retrieved from https://pubmed.ncbi.nlm.nih.gov/33567185/
Williamson, D. A., Bray, G. A., & Ryan, D. H. (2015). Is 5% weight loss a satisfactory criterion to define clinically significant weight loss?. Obesity (Silver Spring, Md.), 23(12), 2319–2320. doi: 10.1002/oby.21358. Retrieved from https://pubmed.ncbi.nlm.nih.gov/26523739/