Key takeaways
Ozempic is an injectable medication for controlling blood sugar in people with type 2 diabetes, along with lifestyle changes. Ozempic is also used to lower the risk of major cardiovascular events in people with diabetes and heart disease and reduce the chance of worsening kidney problems in people with diabetes and chronic kidney disease.
Ozempic comes as an injectable solution in a prefilled pen that should be administered weekly.
After you attach a tiny needle to the pen, you can inject Ozempic under the skin (aka subcutaneously) in one of the three injection sites: the stomach, thigh, or upper arm.
It’s best to rotate Ozempic injection sites every week to prevent irritation, bruising, and other skin reactions.
Here's what we'll cover
Key takeaways
Ozempic is an injectable medication for controlling blood sugar in people with type 2 diabetes, along with lifestyle changes. Ozempic is also used to lower the risk of major cardiovascular events in people with diabetes and heart disease and reduce the chance of worsening kidney problems in people with diabetes and chronic kidney disease.
Ozempic comes as an injectable solution in a prefilled pen that should be administered weekly.
After you attach a tiny needle to the pen, you can inject Ozempic under the skin (aka subcutaneously) in one of the three injection sites: the stomach, thigh, or upper arm.
It’s best to rotate Ozempic injection sites every week to prevent irritation, bruising, and other skin reactions.
If you’ve recently been prescribed Ozempic (semaglutide) or you’re thinking about asking your healthcare provider if it’s right for you, you may be wondering about how and where to inject Ozempic.
It’s common to feel a little nervous about self-injections, but Ozempic—a prescription drug that’s approved by the US Food and Drug Administration (FDA) for controlling blood sugar in people with type 2 diabetes, among other uses, and often prescribed off-label for weight loss—comes with tiny needles (4 mm long) that most people barely feel. To make matters easier, there are only three recommended Ozempic injection sites: the abdomen, thigh, and upper arm. Point being: Once you understand the basics of how to inject Ozempic, giving yourself a weekly shot will feel like second nature.
Keep reading to find out more about injecting Ozempic, from choosing the best place to inject the drug to what to expect when it comes to possible injection site reactions.
Ozempic injection sites: where to inject Ozempic
The best injection sites for Ozempic include the stomach, thigh, or upper arm, according to the drug’s manufacturer Novo Nordisk. Ozempic should be injected subcutaneously (under the skin) and never into a muscle or vein. The skin should be healthy and free of damage, scarring, or injury.
Ozempic comes in prefilled injection pens intended to be self-injected by patients into one of the recommended injection sites once a week. With each injection, you’ll attach a new needle to the pen to administer the injection. The needle itself is quite thin and short—about the width of two human hairs. Subcutaneous injections with such tiny needles tend to be relatively painless.
With each weekly injection, you should use a different site from the prior week. If you choose the same area, be sure to use a different spot in that area. Injecting the same exact spot over and over can lead to lumps or thickened areas under the skin, which could affect how well the medication works. Rotating where you inject Ozempic may also help prevent bruising and irritation at the injection site.
Here’s a bit more detail about the injection sites for Ozempic:
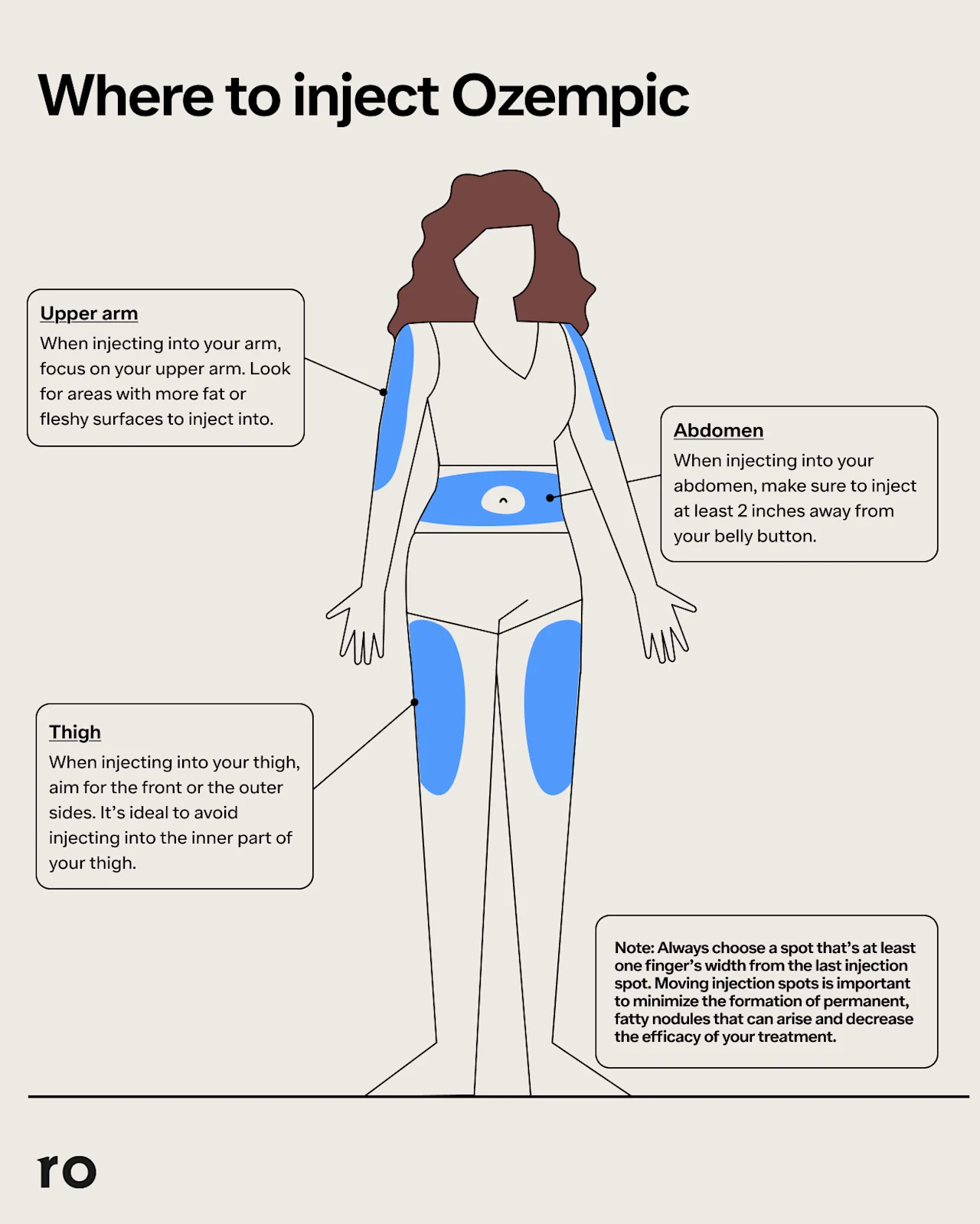
Stomach: The lower abdomen is one of the easiest injection sites for Ozempic because it’s fairly accessible and it has a good amount of subcutaneous fat, which can ensure proper absorption of the medication. When injecting Ozempic in the stomach, choose a spot at least 2 inches away from your belly button and avoid your waistline to prevent clothing rubbing against the injection site and creating irritation.
Thigh: If you choose your thigh as an Ozempic injection site, select a spot on the outer side of your upper thigh, roughly halfway between your knee and hip. Avoid injecting in the inner thigh so your clothing or thighs don’t rub against the spot and cause irritation.
Upper arm: Specifically, this Ozempic injection site refers to the side or back of the upper arm, which is why it can be helpful to have a friend or family member perform this injection for you. Just make sure they choose a spot that’s at least 3 inches below your shoulder and 3 inches above your elbow.
If you give yourself insulin shots, you can inject Ozempic in the same part of your body you use for insulin, such as your abdomen. Just don’t inject them right next to each other (give those injections a little room to breathe!).
Where is the best place to inject Ozempic in general?
The best place to inject Ozempic is under the skin of your upper thigh, abdomen, or upper arm. These are the standard sites for subcutaneous injections in general. Your body absorbs the medication just as well whether you inject it into your abdomen, thigh, or upper arm—so, all three injection sites are equally effective.
Ultimately, the best place to inject Ozempic typically comes down to personal preference. Some people prefer to inject the medication in a less visible area (such as the upper thigh or abdomen) in case of minor skin reactions. Some people, on the other hand, find the abdomen to be a less painful injection site than the thigh.
Where is the best place to inject Ozempic for weight loss?
The best place to inject Ozempic for weight loss is the upper thigh, abdomen, or upper arm. The same three Ozempic injection sites are used, regardless of why you’re taking Ozempic. Any one of these sites can effectively absorb the medication, so the best place to inject it is whichever one is the easiest and most comfortable for you.
Does where you inject Ozempic matter?
Yes, it does. It’s important to stick to the recommended areas: the abdomen, thigh, or upper arm. But you’ll get the same amount of Ozempic in your body, regardless of which of these areas you use—that is, of course, as long as you also follow the instructions included in the drug’s prescribing information and provided by your healthcare provider.
Novo Nordisk recommends those sites likely because you tend to have more layers of fat under the skin in those areas. This makes them ideal locations for a shot that needs to be injected just under the skin, as opposed to an intramuscular injection that needs to go past layers of skin and fat to get deep into a muscle.
How often should you rotate Ozempic injection sites?
You should rotate Ozempic injection sites weekly to avoid irritation or infections. That gives you up to five different injection sites (e.g. right upper arm, left upper arm, abdomen, right upper thigh, left upper thigh). Consider making a note of the date and specific injection site to help you keep track, since it can be tough to remember a week later.
If you find you prefer certain injection sites over others (or if you don’t have someone who can give you the injection in your arm), you can use the same general area—just be sure to inject in a spot at least 1 inch away from where you injected Ozempic last time. This will give the skin time to heal and enable your body to effectively absorb the medication.
Ozempic injection sites diagram
How to inject Ozempic
Ozempic should be self-injected weekly, on the same day each week. So, for example, if you take your first dose on a Sunday, you may want to set yourself a reminder to inject every Sunday.
Your healthcare provider can show you how to use your Ozempic pen the first time. You should also read the manufacturer’s instructions that come with your medication before your first injection. Here’s how to inject Ozempic:
Step 1: Gather your supplies
Before injecting Ozempic, make sure you have all the necessary supplies:
An Ozempic pen and a new pen needle
An alcohol swab or soap and water
A gauze pad or cotton ball
A sharps disposal container
Read the label on your pen and confirm that the medication has not expired. Also, confirm that you have the correct dose of Ozempic. The labels on the pens are color-coded to make this easier:
0.25 mg, 0.5 mg: red
1 mg: teal
2 mg: yellow
Next, look at the medicine in your pen. It should be clear and colorless. If you notice any particles or cloudiness, do not use the pen and contact your pharmacy.
Step 2: Wash your hands
Wash your hands with soap and water, and dry them thoroughly with a clean towel.
Step 3: Prepare your Ozempic pen
Remove the outer cap and attach a new needle to your Ozempic pen. Keep the cap on the needle while you push it onto the pen and turn until it’s on tight. Always use a new needle for each injection to help prevent issues like infections or clogged needles.
When you’re ready to go to the next step, remove the inner and outer needle cap.
Step 4: Prepare your dose of Ozempic
Dial the dose counter so the desired dose lines up with the dose pointer. Follow the dosing instructions from your provider to select the correct dose.
However, if this is your first time using a new pen, you’ll first need to check the Ozempic flow, as instructed below. Otherwise, proceed to Step #4.
Dial the dose selector to the flow check symbol. With the needle pointing up, press and hold the dose button until it goes to 0. You should see a drop at the tip of the needle.
If you don’t see a drop, repeat up to six more times until you do. If you still don’t see a drop at the tip of the needle, use a new pen or contact Novo Nordisk at (888) 693-6742.
Step 5: Inject your dose of Ozempic
Use an alcohol swab to clean the skin where you will inject. Let your skin air dry.
Position the pen so you can see the dose counter, and insert the Ozempic needle into your skin at a 90-degree angle into the injection site (belly, thigh, or upper arm).
Press and hold down the dose button until 0 mg lines up with the dose pointer. With the button still pressed and the needle still inserted, slowly count for six seconds to allow a full dose of Ozempic to inject. Keeping your thumb on the dose button, remove the needle from your skin.
Step 6: Dispose of your needle and store your pen
Remove the needle from the pen and throw it away in an FDA-approved sharps disposal container. You can also dispose of used Ozempic pens in the sharps container. Do not toss either in the household garbage or recycling.
You can get an official sharps container from a pharmacy or medical supply retailer. You can also use a heavy-duty plastic container as an alternative—just make sure it meets the FDA requirements for a sharps disposal container:
Made of heavy-duty plastic
Able to close with a tight-fitting, puncture-proof lid
Stable when propped upright
Leak-resistant
Properly labeled (i.e. clearly marked with a warning of the hazardous waste)
Put the pen cap back on your pen and store it at room temperature, away from light, until your next dose. After you’ve opened an Ozempic pen, it can be stored at room temperature (59°F–86°F) or in a refrigerator (36°F–46°F). Before your first use of the pen, Ozempic should be stored in the refrigerator.

Ozempic injection site reactions
Ozempic injection site reactions are relatively rare. In placebo-controlled trials, only 0.2% of the participants using Ozempic experienced injection site reactions. These included discomfort or redness where they injected the medication.
When you remove the needle after injecting Ozempic, you may notice some blood at the injection site. This is nothing to be concerned about; simply press on it gently with clean gauze.
If you have an allergic reaction to Ozempic, you may experience swelling at the injection site or elsewhere, along with trouble breathing, rash, dizziness, fainting, or a fast heartbeat. If you believe you are having an allergic reaction, seek medical attention immediately.
Do Ozempic injections hurt?
Generally, Ozempic injections aren’t very painful because they are subcutaneous injections that go just below the surface of the skin using a thin, short needle (4 mm long, which is about one-sixth of an inch). (In comparison, many vaccines and other shots that get injected deeper into the muscle are given using a needle that is 1–1.5 inches long.)
Ozempic injection site pain is relatively rare, affecting less than 1% of people in clinical trials. And that number includes injection site reactions outside of pain, such as rash or general discomfort.
How to make sure you get your full Ozempic dose
To make sure you get your full Ozempic dose, follow the instructions included with the medication and any other guidance you’ve received from your healthcare provider. Once you’ve inserted the needle into your skin, press and hold down the dose button. Wait for the dose counter to reach 0 mg, and then slowly count to six before removing the needle from your skin. If you follow these instructions, you can rest assured that you’ve likely administered your entire dose of Ozempic.
Here are some more tips to follow to ensure you get your full dose of Ozempic every time:
Before each injection, inspect your pen to ensure that you have the right dose, it hasn’t expired, and the medication is clear of any particles or cloudiness.
Inject Ozempic into healthy skin that’s free of injuries, swelling, redness, scarring, bruising, or other damage.
Inject your dose into one of the three recommended Ozempic injection sites—upper thigh, abdomen, or upper arm—and rotate through these areas with each injection.
Insert the needle into your skin at a 90-degree angle, pressing down firmly until the dose counter reaches 0 and you’ve counted for a full six seconds.
If you’re unsure if you’ve delivered your full dose of Ozempic or have other questions about properly injecting the medication, contact your healthcare provider or pharmacist. They can answer your questions and demonstrate how to use the Ozempic pen at the appropriate injection sites.
Bottom line
If you’re new to Ozempic, you might feel a little unsure about how or where to inject it. But once you get the hang of it, administering the medication becomes a quick and simple part of your weekly routine. Injecting Ozempic is easy and relatively painless. You’ll inject it just under your skin into one of three injection sites: your stomach, upper thigh, or upper arm.
Choose a healthy injection site. Inject Ozempic in the abdomen, upper thigh, or upper arm. Avoid areas with bruising, redness, or scarring.
Rotate through the Ozempic injection sites each week. Don’t inject into the same exact spot two weeks in a row. This helps prevent irritation and ensures better medication absorption.
Inject Ozempic at the correct angle. Insert the needle straight into your skin at a 90-degree angle, press the dose button until it reaches 0, and slowly count to six before you remove the needle.
Check your Ozempic pen before you use it. Make sure the medication is clear and colorless, hasn’t expired, and that you have the correct dose.
Ask for help if needed. If you’re unsure how to inject Ozempic or can’t reach certain spots—like the back of your arm—ask a friend, family member, or healthcare provider for help.
Frequently asked questions (FAQs)
Can you inject Ozempic too deep?
The needles that come with the Ozempic pen are very short and thin, so you shouldn’t be able to inject Ozempic “too deep,” as long as you follow the medication’s instructions. This means using one of the three Ozempic injection sites (upper thigh, abdomen, upper arm) and injecting into the fatty tissue just under the skin. Never inject Ozempic into a muscle or vein.
Why do you get a lump after injecting Ozempic?
It’s possible to get a lump or bump under your skin after injecting Ozempic. If you notice this, it’s a sign that you aren’t rotating the injection site or using a new needle for each injection. These lumps (technically known as lipohypertrophy) can develop when you don’t rotate through the injection sites or when you don’t inject directly into the fatty tissue, as recommended. Avoid injecting Ozempic into a lumpy or scarred area of your skin because the medication may not absorb properly.
Where is the least painful injection site for Ozempic?
Some people find the abdomen to be a less painful injection site than the thigh, according to research on subcutaneous injections like Ozempic.
Is it better to inject Ozempic in the thigh or stomach?
Both the thigh and stomach (along with the upper arm) are recommended injection sites for Ozempic. Whichever is “better” is a matter of personal preference. However, some research shows that people generally find the stomach to cause less Ozempic injection site pain than the thigh.
How far from the belly button should you inject Ozempic?
When injecting Ozempic in the stomach, you should choose a spot that is at least 2 inches away from your belly button. Select a spot on the lower abdomen that’s below your ribs but above your hip bones.
How do I know if I did my Ozempic injection correctly?
If you follow the instructions included with your Ozempic pen, you can probably rest assured that you’ve done your Ozempic injection correctly. But here are some telltale signs that you properly injected Ozempic:
You watched the dose counter reach 0 mg.
You held the Ozempic needle in place for at least six seconds afterward.
You didn’t see any medication leak out when you removed the needle from your skin.
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
Ozempic Important Safety Information: Read more about serious warnings and safety info.
References
Canadian Agency for Drugs and Technologies in Health (CADTH). (2019). Clinical Review Report: Semaglutide (Ozempic): (Novo Nordisk Canada Inc.). Canadian Agency for Drugs and Technologies in Health. Retrieved from https://pubmed.ncbi.nlm.nih.gov/31305971/
DailyMed. (2025). Label: Ozempic- semaglutide injection, solution. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=adec4fd2-6858-4c99-91d4-531f5f2a2d79
Hearn, E. B. & Sherman, J. J. (2021). Injection-site nodules associated with once-weekly subcutaneous administration of semaglutide. Diabetes Spectrum: A Publication of the American Diabetes Association, 34(1), 73–75. doi: 10.2337/ds20-0033. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC7887535/
Heise, T., Nosek, L., Dellweg, S., et al. (2014). Impact of injection speed and volume on perceived pain during subcutaneous injections into the abdomen and thigh: a single-centre, randomized controlled trial. Diabetes, Obesity & Metabolism, 16(10), 971–976. doi: 10.1111/dom.12304. Retrieved from https://pubmed.ncbi.nlm.nih.gov/24720741/
Kommu, S. & Whitfield, P. (2024). Semaglutide. StatPearls. Retrieved on Feb. 4, 2025 from https://www.ncbi.nlm.nih.gov/books/NBK603723/
MedlinePlus. (2024). Semaglutide injection. American Society of Health-System Pharmacists, Inc. Retrieved from https://medlineplus.gov/druginfo/meds/a618008.html
MedlinePlus. (2023). Subcutaneous (SQ) injections. Retrieved from https://medlineplus.gov/ency/patientinstructions/000430.htm
Novo Nordisk. (2024). Using your Ozempic pen. Once-Weekly Ozempic Subcutaneous Injection. Retrieved on Feb. 4, 2025 from https://www.ozempic.com/how-to-take/ozempic-pen.html
Tian, T., Aaron, R. E., Huang, J., et al. (2023). Lipohypertrophy and Insulin: An Update From the Diabetes Technology Society. Journal of Diabetes Science and Technology, 17(6), 1711–1721. doi: 10.1177/19322968231187661. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC10658672/
U.S. Food and Drug Administration (FDA). (2025). Ozempic (semaglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl.pdf
Usach, I., Martinez, R., Festini, T., & Peris, J. E. (2019). Subcutaneous injection of drugs: literature review of factors influencing pain sensation at the injection site. Advances in Therapy, 36(11), 2986–2996. doi: 10.1007/s12325-019-01101-6. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC6822791/