Key takeaways
The starting dosage of Mounjaro is 2.5 mg once weekly, and the maximum weekly dose is 15 mg.
Every four weeks, your healthcare provider may increase the dose until you reach a maintenance dose that is effective for your health needs.
For some people with type 2 diabetes, a 5 mg dose may be enough to control blood sugar levels, while others will require a higher dose.
Higher doses of Mounjaro lead to more weight loss but may also have more side effects.
Here's what we'll cover
Key takeaways
The starting dosage of Mounjaro is 2.5 mg once weekly, and the maximum weekly dose is 15 mg.
Every four weeks, your healthcare provider may increase the dose until you reach a maintenance dose that is effective for your health needs.
For some people with type 2 diabetes, a 5 mg dose may be enough to control blood sugar levels, while others will require a higher dose.
Higher doses of Mounjaro lead to more weight loss but may also have more side effects.
If you have type 2 diabetes, obesity, or both, your healthcare provider may recommend taking Mounjaro (tirzepatide). This once-weekly injectable prescription medication follows a ramp-up dosage schedule, wherein your healthcare provider will gradually increase your Mounjaro dosage until finding an effective maintenance dose that controls your blood sugar levels or produces weight loss (or both).
If this is your first time taking Mounjaro, you may have questions about the Mounjaro dosage for weight loss or type 2 diabetes. Below we share what you need to know about Mounjaro dosing, including how to use Mounjaro and what to do if you miss a dose. Read on.
What is the typical Mounjaro dosage for weight loss?
The typical Mounjaro dosage for weight loss is 5 mg, 10 mg, or 15 mg weekly.
Mounjaro comes in six dosage strengths, ranging from 2.5 mg (starting or lowest dose) to 15 mg (maximum maintenance dose). Usually, healthcare providers prescribe a Mounjaro starting dose of 2.5 mg to help your body get used to the medication. Then, after four weeks, they may increase your Mounjaro dose to 5 mg. Moving forward, your provider may continue increasing your dose in increments of 2.5 mg every four weeks or so, depending on your tolerance of side effects and the weight loss results you experience. This gradual dose escalation is designed to keep side effects manageable until you find the right maintenance dosage for you, be it 5 mg, 10 mg, or 15 mg weekly.
Note: Mounjaro is approved by the US Food and Drug Administration (FDA) for type 2 diabetes, not weight loss. But it contains the same active ingredient—and the same dosage strengths—as Zepbound, which is FDA-approved for weight loss and management as well as sleep apnea. These aforementioned dosage guidelines for weight loss are based on Zepbound’s prescribing information, which healthcare providers may reference when prescribing Mounjaro off-label for weight loss.
What is Mounjaro’s dosage?
The recommended starting dose of Mounjaro is 2.5 mg, taken weekly. The maximum dosage is 15 mg weekly. It comes in single-dose pens (and single-dose vials outside of the US), which you’ll use to inject the solution under the skin of your upper arm, abdomen, or thigh. (More on this below.)
When you first start taking Mounjaro, your healthcare provider will likely start you at the typical starting dosage of 2.5 mg weekly. After four weeks, they may increase your dosage to 5 mg weekly. After another four weeks, they’ll check how the medication is affecting your blood sugar levels. If the 5 mg dosage adequately improves your glycemic control, your healthcare provider may have you stay at this dosage long-term. If your blood sugar is still high, they may continue increasing your dose of Mounjaro by 2.5 mg every four weeks until they find the right Mounjaro maintenance dose for you.
For example, if your blood sugar levels remain high after four weeks of the 7.5 mg dosage, your healthcare provider may increase your dose to 10 mg. If your blood sugar levels look good after another four weeks at the 10 mg dose, they may have you stay at that dosage. If they’re still too high, they can keep increasing it every four weeks until the maximum dosage of 15 mg. The goal is to find the lowest dosage of Mounjaro that adequately controls your blood sugar levels without significant side effects.
Mounjaro dosage strengths
Each dose of Mounjaro comes in a single-dose pen. There are six strengths of Mounjaro:
2.5 mg/0.5 mL
5 mg/0.5 mL
7.5 mg/0.5 mL
10 mg/0.5 mL
12.5 mg/0.5 mL
15 mg/0.5 mL
Mounjaro dosage schedule
Healthcare providers typically prescribe the lowest Mounjaro starting dose of 2.5 mg. Then, depending on your response to the medication, they may gradually increase your Mounjaro dosage every four weeks until they find the right maintenance dose for you. Dosage increases usually occur in 2.5-mg increments in order to allow your body to get used to the medication and keep common side effects—namely, nausea, diarrhea, and vomiting—manageable. The following Mounjaro dosage chart shows a typical dose escalation schedule:
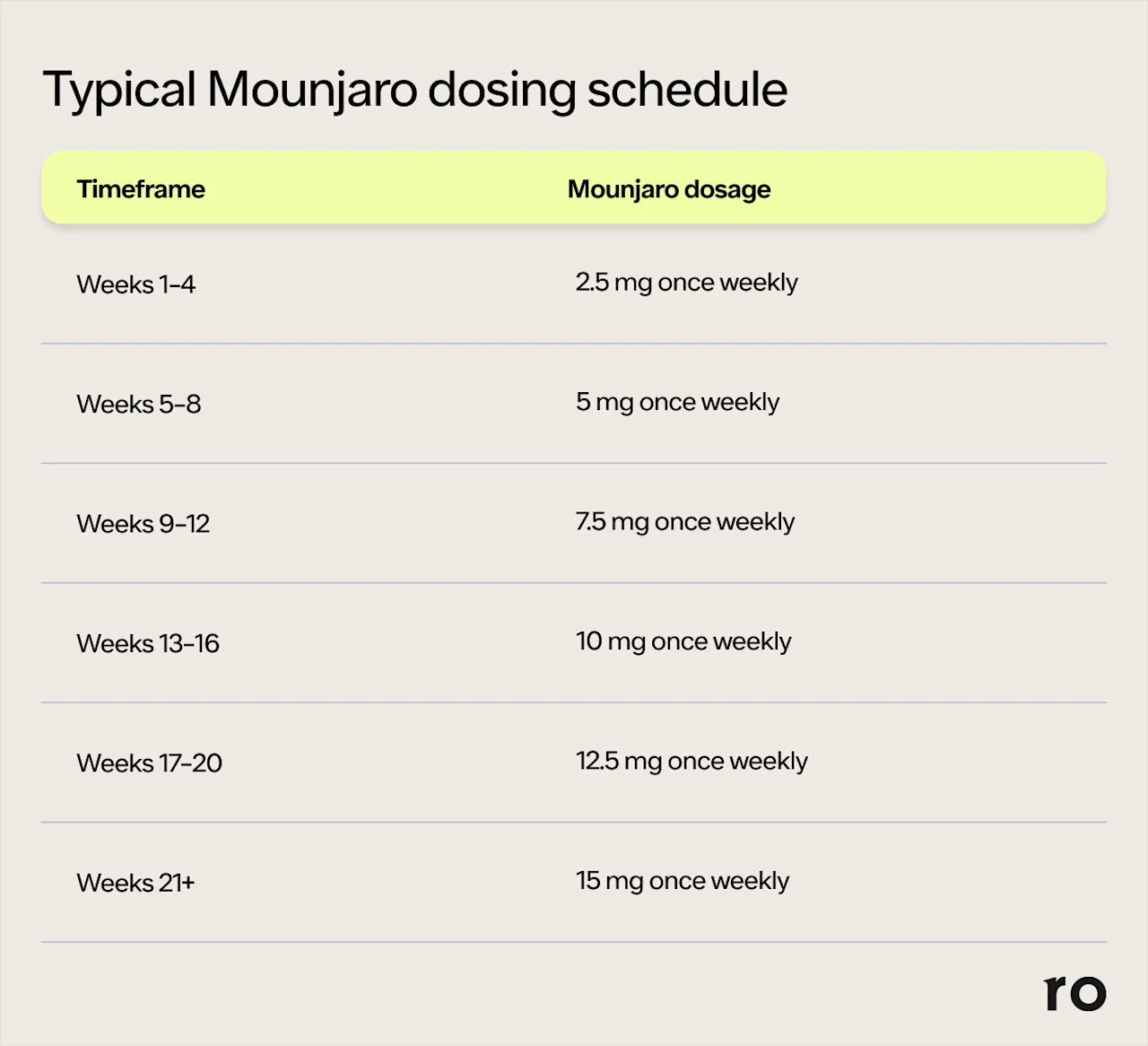
*The dose adjustments for Mounjaro typically occur every 4 weeks, but your healthcare provider may titrate you more slowly or more quickly, depending on your response to the medication. If you titrate up to a higher dose and experience side effects that become too much, they may take you back down to a lower dose. Keep in mind that many patients see results with just 5 mg and, therefore, may not have to continue to increase their dose.
As you can see, it can take 1–4 months to reach your maintenance dose of Mounjaro. But it’s worth noting that not everyone will follow this exact schedule or even need to take the highest dose of Mounjaro. Some people may find that a lower dose of 5 mg or 10 mg adequately controls their blood sugar levels, and some people may transition more slowly between the different strengths. If you have trouble tolerating a dose increase, your healthcare provider may have you go back down to the previous Mounjaro dose. That’s just one of the many reasons you should continue to stay in communication with your provider throughout treatment.
The goal is to find the lowest effective dose of Mounjaro that produces the desired therapeutic effect(s) for you, be it glycemic control or weight loss, and keeps side effects manageable so you can continue taking the medication.
What is the Mounjaro starting dose?
The Mounjaro starting dose is 2.5 mg, injected weekly. This starting dose is only recommended for the first four weeks to reduce the risk of side effects like nausea, diarrhea, and vomiting. It is not FDA-approved as a long-term treatment for blood sugar control. After four weeks, your healthcare provider will likely bump you up to 5 mg weekly unless you have significant side effects.
What is the highest dose of Mounjaro?
The highest dose of Mounjaro is 15 mg, injected weekly. Although, your healthcare provider may keep you at a lower maintenance dose of 5 mg or 10 mg if that adequately improves your blood sugar levels (or produces weight loss, if you’re taking Mounjaro off-label for that purpose). If this dose of Mounjaro is safe and effective for you, your healthcare provider will likely recommend you continue taking it long-term.
What is the Mounjaro maintenance dose?
There are three Mounjaro maintenance doses: 5 mg, 10 mg, or 15 mg. The other doses of Mounjaro—2.5 mg, 7.5 mg, and 12.5 mg—are intended to be taken as transitional doses in between the maintenance doses so your body can gradually adjust to the medication and its side effects. Remaining on 2.5 mg, 7.5 mg, or 12.5 mg for longer than one month is considered an off-label prescribing practice that is used to help people develop even more gradual tolerance to the medication’s side effects. Once you find a Mounjaro maintenance dose that is safe and effective for you, your healthcare provider will likely recommend you continue taking it long-term.
What factors affect Mounjaro dose?
Different people may be prescribed different dosages of Mounjaro. For example, some people may require higher doses of Mounjaro to adequately control their blood sugar levels. To allow healthcare providers to provide the best dosage, Mounjaro is available in six strengths, from 2.5 mg to 15 mg. The dose of Mounjaro your healthcare provider prescribes for you can depend on several factors, including your age, insurance, and personal medical history.
Your age
Mounjaro has not been approved for people younger than 18 years of age. Across seven clinical trials, about one in three people taking Mounjaro were 65 or older.
While clinical trials have not found any significant differences in how Mounjaro affects older adults vs. those younger than 65 years of age, some older adults may be more sensitive to Mounjaro side effects. That said, the starting dosage and dosing schedule for Mounjaro is typically the same for adults regardless of age.
Other medical conditions
Some medications come with different dosage recommendations for people with kidney or liver problems, but Mounjaro isn’t one of them.
Still, healthcare professionals will use more caution when prescribing Mounjaro to people with certain medical conditions, such as a history of diabetic retinopathy (diabetes-related vision loss or changes), pancreatitis, severe gastrointestinal disease, acute kidney injury, or acute gallbladder disease. Given their medical history, these people may be more likely to experience adverse reactions when using Mounjaro.
People with a personal or family history of thyroid cancer, including multiple endocrine neoplasia syndrome type 2 (MEN 2) or medullary thyroid carcinoma (MTC), should not use Mounjaro.
The condition for which you’re taking Mounjaro
Mounjaro is available in six strengths ranging from 2.5 mg to 15 mg. Most people begin treatment with the starting dosage of 2.5 mg for four weeks before increasing to 5 mg. Your healthcare provider may continue increasing your dose depending on the condition you are taking Mounjaro for—type 2 diabetes mellitus or for weight loss—and the severity of that condition.
For some people with type 2 diabetes, a 5 mg dose can be sufficient to control their blood sugar levels, while others will require a higher dosage of Mounjaro. Higher doses of Mounjaro lead to more weight loss but may also have more side effects.
Other medications you’re taking, particularly those that lower blood sugar levels
Because Mounjaro lowers blood glucose, it may increase the risk of hypoglycemia for people who take other medications that lower blood glucose, like insulin or similar medicines. To reduce your risk, your healthcare provider may decrease your current dose of insulin.
While Mounjaro has no other known drug interactions, it does delay gastric emptying, so it may affect the rate at which your body absorbs other medications you take orally. Be sure to tell your healthcare provider about any medications you are taking prior to starting Mounjaro, so they can advise you if you need to make any changes.
For example, if you take birth control pills, they may recommend switching to a non-oral form of contraception or using condoms as a backup method for four weeks after starting Mounjaro and after each dose increase. This is important because it’s possible that the medication may reduce the efficacy of birth control. Tell your healthcare provider if you become pregnant or plan to become pregnant.
How to use Mounjaro
Mounjaro should be injected subcutaneously (under the skin) every week. You can inject Mounjaro at any time of day, regardless of when you last ate. Store Mounjaro in its original carton in your refrigerator at a temperature between 36°F to 46°F (2°C to 8°C). Do not freeze Mounjaro. You can also store Mounjaro at room temperature, up to 86°F (30°C), for up to 21 days.
Each box of Mounjaro contains detailed prescribing information for injecting and storing Mounjaro, including illustrations for each step we describe below. Read through the paperwork and ask your healthcare provider (or pharmacist) if you have any questions. You can also watch a video on the manufacturer’s website or ask your healthcare provider to show you or your caregiver how to inject Mounjaro.
While you should follow your healthcare provider’s instructions, here is a general overview of what you can expect.
Step 1: Choose your injection site
Mounjaro is injected in the abdomen, thigh, or upper arm. If you choose your upper arm as your spot, it’s best to have another person or caregiver do so to ensure the medicine gets injected easily. Change your injection site each week, rotating through the different areas of the body.
If you want to continue injecting in the same area, you can, but don’t inject Mounjaro in the exact same spot. If you also take insulin, you can inject Mounjaro and insulin in the same general area of your body (e.g. abdomen), but they should not be injected right next to each other or mixed together into the same injection.
Step 2: Prepare the medication
If you are injecting Mounjaro using a pen, make sure the pen is locked. Then, pull the gray base cap off the pen and throw it away. Do not touch the needle. Place the clear base of the pen so it lies flat against the skin of your injection site. Unlock the pen by turning the lock ring.
Step 3: Inject Mounjaro
Press the purple injection button and hold for 10 seconds. Listen for two clicks. The first click indicates that the injection has started, while the second click indicates that it has completed. The injection is complete when the gray plunger becomes visible.
Step 4: Safely dispose of the used medication
Used Mounjaro pens and needles should only be disposed of in an FDA-approved sharps disposal container (a hard plastic container designed for needles). Do not put the pens in your household trash.
Missed Mounjaro dose
What should you do if you forget to take a dose of Mounjaro? It depends on the timing of your last dose.
If it has been less than four days (96 hours) since the day you were supposed to take your dose, go ahead and inject Mounjaro as soon as you remember. Then, continue with your weekly dosing schedule as usual.
If it has been more than four days, though, you should skip that dose of Mounjaro and just take the next dose on your regularly scheduled day.
Never take two doses of Mounjaro at the same time. If you have any questions about a missed dose, call your healthcare provider. You can also consult your pharmacist.
If you skip too many doses of Mounjaro, you may need to lower your dosage or restart treatment at 5 mg to minimize side effects. Your healthcare provider can advise you on the next steps if you miss multiple doses.
If you need to change the day you inject Mounjaro, that’s doable. The key is to make sure you don’t take two doses within 72 hours of each other. Again, you can contact your healthcare provider if you have any questions or concerns.
Missed Mounjaro dosage chart
If you miss a dose of Mounjaro, here’s an example of what to do:

Do you have to take Mounjaro on the same day?
Yes, Mounjaro should be taken weekly, on the same day each week. This dosing schedule is based on the drug’s half-life. When you start Mounjaro, choose a day of the week that should work consistently for you. To prevent missing a dose, you might choose a day you reliably do the same thing, such as laundry or grocery shopping. It’s also a good idea to set a reminder on your phone or make a note on your calendar. If you need to change the day you take your Mounjaro dose, you can do so as long as it has been at least three days (72 hours) since your last dose.
Can you change your Mounjaro injection day?
Mounjaro works best when you take it consistently, on the same day each week. If you need to change your Mounjaro injection day, you can do so as long as at least three days (72 hours) have passed since your last dose. For example, if you currently take Mounjaro on Saturday mornings and want to switch to Wednesday, you would take your dose this Saturday, then start your new schedule on Wednesday. Be sure to update any reminders on your phone or calendar to your new day.
What happens if you take too much Mounjaro?
You should avoid taking two doses of Mounjaro within three days (72 hours) of each other. If you accidentally take too much Zepbound, contact America’s Poison Centers at 800-222-1222 or visit their website at poisonhelp.org for next steps. You can also contact your healthcare provider or visit a local Urgent Care or Emergency Department for immediate medical attention. Taking more Mounjaro than prescribed raises the risk of side effects.
The most common Mounjaro side effects are gastrointestinal issues (mainly nausea, diarrhea, vomiting, constipation, and indigestion). In some cases, these side effects can be severe enough to lead to dehydration or even kidney failure. Rarely, other serious side effects have been reported. If side effects don't go away or feel severe, consult a healthcare professional for advice or medical attention.
Are there any Mounjaro dosage adjustments for medical conditions or interactions?
If you are taking certain other medications, those dosages may need to be adjusted to prevent negative interactions with Mounjaro. For example, since Mounjaro lowers blood sugar levels, your risk of experiencing hypoglycemia (low blood sugar) is raised when you combine Mounjaro with other medications that also lower blood sugar, such as insulin. Your healthcare provider may lower your dose of insulin or sulfonylureas to reduce this risk.
Mounjaro also delays gastric emptying (how quickly food moves out of your stomach), which may affect the absorption of any oral medications you’re taking, such as birth control pills. If you’re taking oral contraceptives, your healthcare provider may recommend switching to a non-oral contraceptive method (such as an IUD or implant) or using a barrier method (such as condoms) for four weeks when starting Mounjaro and after each dose increase.
Mounjaro side effects
As with most prescription drugs, you may experience side effects when taking Mounjaro. Fortunately, the side effects tend to be most common when you first start using the drug, and go away with time as your body gets used to Mounjaro. For most people, Mounjaro side effects range from mild to moderate and are typically gastrointestinal in nature. They may include:
Nausea
Diarrhea
Decreased appetite
Vomiting
Constipation
Upset stomach or indigestion
Abdominal pain
Some side effects—including nausea, diarrhea, decreased appetite, vomiting, and constipation—tend to be more common with higher doses of Mounjaro, according to clinical trials of people taking Mounjaro for type 2 diabetes. For example, nausea, diarrhea, and vomiting were reported by 32.7% of people taking 5 mg of Mounjaro, 51% taking 10 mg, and 66% taking 15 mg.
In studies of people with obesity but not type 2 diabetes, gastrointestinal side effects like nausea, diarrhea, and vomiting were also more common among people taking higher doses of tirzepatide (10 mg or 15 mg) vs. those taking 5 mg.
Adults with type 2 diabetes who also take insulin may have a slightly higher risk of experiencing severe hypoglycemia(low blood sugar) with higher doses (10 mg or 15 mg) of tirzepatide, according to another trial. Starting with a low dose and slowly increasing the dose may help make gastrointestinal side effects more manageable—hence why Mounjaro has a gradual dose escalation over the first few months of treatment.
If you experience nausea while taking Mounjaro, Eli Lilly, the makers of Mounjaro recommend the following:
Avoiding fatty foods
Eating foods that are bland and light, such as toast, crackers, or rice
Eating smaller meals
No longer eating once you feel full
Your healthcare provider will prescribe the appropriate dosage of Mounjaro for you. Follow their medical advice and use Mounjaro exactly as prescribed. If you notice any side effects or have any questions about your dosage of Mounjaro, talk to your healthcare provider. They can offer suggestions for managing side effects. They may adjust your dosage or recommend an alternative medication like Ozempic or Wegovy.
Rx weight loss with Ro
Get access to prescription weight loss medication online
Bottom line
Finding the right dosage of Mounjaro is a process that takes time, patience, and medical guidance. Whether you're using Mounjaro for blood sugar control or weight loss, the goal is to find the lowest effective dose that meets your health goals.
Mounjaro comes in six strengths, ranging from 2.5 mg to 15 mg. The Mounjaro starting dose is 2.5 mg weekly, and the maximum dose is 15 mg weekly. Common Mounjaro maintenance doses are 5 mg, 10 mg, or 15 mg weekly.
Mounjaro dose escalation increases typically occur every four weeks in increments of 2.5 mg. The goal is to reach an effective maintenance dose that produces results and keeps side effects manageable.
Nausea, diarrhea, and vomiting are the most common side effects, especially at higher doses. But they often improve over time as your body gets used to the medication.
If you miss a dose by less than four days, take the medication as soon as you remember. If more than four days have passed since you last took Mounjaro, skip the dose and resume your regular schedule. Don’t take extra medication to make up for a missed dose. Mounjaro doses should always be taken at least three days apart.
Frequently asked questions (FAQs)
Can I split my Mounjaro dose?
Mounjaro comes in single-dose pens. Each pen contains a single dose of the medication to ensure accurate dosing. Each dose of Mounjaro should be injected completely once per week.
What should you do about your Mounjaro dosage if you go on vacation?
If you’re going on vacation, bring your Mounjaro with you. While Mounjaro is ideally stored in the refrigerator, it can be stored at room temperature for up to 21 days. Just make sure your Mounjaro is stored in an area that does not exceed 86°F (30°C). Keep your doses in their original labeled carton in case security needs to screen them. This will also protect your Mounjaro dose from light.
If you improve your diet and exercise routine, will your Mounjaro dosage be lowered?
When taking Mounjaro, you should implement lifestyle changes like improving your diet and exercise routine. Making these changes can significantly improve blood sugar levels in people with type 2 diabetes. When taking Mounjaro, your healthcare provider should routinely monitor your HbA1c levels to determine if your Mounjaro dosage should change.
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
Mounjaro Important Safety Information: Read more about serious warnings and safety info.
Zepbound Important Safety Information: Read more about serious warnings and safety info.
Ozempic Important Safety Information: Read more about serious warnings and safety info.
Wegovy Important Safety Information: Read more about serious warnings and safety info.
References
Chavda, V. P., Ajabiya, J., Teli, D., et al. (2022). Tirzepatide, a New Era of Dual-Targeted Treatment for Diabetes and Obesity: A Mini-Review. Molecules (Basel, Switzerland), 27(13), 4315. doi: 10.3390/molecules27134315. Retrieved from https://pubmed.ncbi.nlm.nih.gov/35807558/
Farzam, K. & Patel, P. (2024). Tirzepatide. StatPearls. Retrieved from https://pubmed.ncbi.nlm.nih.gov/36251836/
García-Molina, L., Lewis-Mikhael, A. M., Riquelme-Gallego, B., et al. (2020). Improving type 2 diabetes mellitus glycaemic control through lifestyle modification implementing diet intervention: a systematic review and meta-analysis. European Journal of Nutrition, 59(4), 1313–1328. doi: 10.1007/s00394-019-02147-6. Retrieved from https://pubmed.ncbi.nlm.nih.gov/31781857/
Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. The New England Journal of Medicine, 387(3), 205–216. doi: 10.1056/NEJMoa2206038. Retrieved from https://pubmed.ncbi.nlm.nih.gov/35658024/
Lilly. (2024). How to Use, Dosing & Side Effects | Mounjaro® (tirzepatide). Retrieved from https://www.mounjaro.com/how-to-use-mounjaro
Min, T. & Bain, S. C. (2021). The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Therapy: Research, Treatment and Education of Diabetes and Related Disorders, 12(1), 143–157. doi: 10.1007/s13300-020-00981-0. Retrieved from https://pubmed.ncbi.nlm.nih.gov/33325008/
Mishra, R., Raj, R., Elshimy, G., et al. (2023). Adverse Events Related to Tirzepatide. Journal of the Endocrine Society, 7(4), bvad016. doi: 10.1210/jendso/bvad016. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC9915969/
U.S. Food and Drug Administration (FDA-a). (2024). Highlights of Prescribing Information: Mounjaro (tirzepatide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215866s010s015s022lbl.pdf
U.S. Food and Drug Administration (FDA-b). (2024). Highlights of Prescribing Information: Zepbound (tirzepatide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/217806s013lbl.pdf
Whitley, H. P., Trujillo, J. M., & Neumiller, J. J. (2023). Special Report: Potential Strategies for Addressing GLP-1 and Dual GLP-1/GIP Receptor Agonist Shortages. Clinical Diabetes: A Publication of the American Diabetes Association, 41(3), 467–473. doi: 10.2337/cd23-0023. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC10338283/