Key takeaways
If there’s a drug shortage or your insurance doesn’t cover Ozempic, other medications are available that might be good for weight loss or type 2 diabetes.
Ozempic alternatives include other GLP-1 agonists, such as those that are FDA-approved to treat type 2 diabetes (e.g. Mounjaro, Trulicity, Rybelsus) and for chronic weight management (e.g. Wegovy, Zepbound).
Oral medications, such as metformin, Qsymia, Contrave, and Xenical may also be prescribed as Ozempic alternatives, though they have different ways of working.
Here's what we'll cover
Key takeaways
If there’s a drug shortage or your insurance doesn’t cover Ozempic, other medications are available that might be good for weight loss or type 2 diabetes.
Ozempic alternatives include other GLP-1 agonists, such as those that are FDA-approved to treat type 2 diabetes (e.g. Mounjaro, Trulicity, Rybelsus) and for chronic weight management (e.g. Wegovy, Zepbound).
Oral medications, such as metformin, Qsymia, Contrave, and Xenical may also be prescribed as Ozempic alternatives, though they have different ways of working.
Maybe there’s a drug shortage. Or perhaps your insurance doesn’t cover it. Whatever the case may be, if you’re considering Ozempic (semaglutide), you may be wondering if there are any Ozempic alternatives that can also help manage blood sugar and encourage weight loss, among other effects.
The good news is that Ozempic is not the only medication in its class. There are several other drugs that are similar to Ozempic and can produce similar effects, especially when combined with a healthy diet and exercise.
It’s important to note, however, that most of the Ozempic alternatives are, like Ozempic, approved by the US Food and Drug Administration (FDA) to control blood sugar in those with type 2 diabetes. And while a few have received FDA approval for chronic weight management, others may be prescribed off-label for weight loss.
Ahead, we break down nine Ozempic alternatives, including options for diabetes and weight loss. But remember: When considering medications, it’s best to consult a healthcare practitioner. So, think of this dive into Ozempic alternatives as a prep course, if you will, for a forthcoming discussion on your best option(s) with your provider.
9 Ozempic alternatives
As mentioned above, many Ozempic alternatives are also glucagon-like peptide-1 (GLP-1) receptor agonists. These medications work by directing the pancreas to release more insulin when blood glucose levels get too high and limiting the release of glucagon, a hormone that raises your blood glucose levels. Together, these effects are what make GLP-1s so effective at stabilizing blood glucose in people with type 2 diabetes.
GLP-1 receptor agonists also slow down digestion, leaving you feeling fuller longer, which may lead you to eat less food and experience weight loss. In addition to decreasing appetite, GLP-1s also tell the brain you’re full after eating, which adds another layer of appetite regulation. It’s because of these impacts that Ozempic and many of its alternatives are often prescribed off-label for weight loss.
Since several of the Ozempic alternatives in this article work in similar ways, they tend to share side effects with Ozempic, including nausea, vomiting, abdominal pain, diarrhea or constipation, and injection site reactions (e.g. pain or skin irritation). In general, these symptoms are most common when you start using the medications and as you increase your dose.
And one more point of note before diving into the Ozempic alternatives: All of the options below work best when used in tandem with lifestyle changes, such as following a balanced diet and regular exercise.
Ozempic alternatives for type 2 diabetes
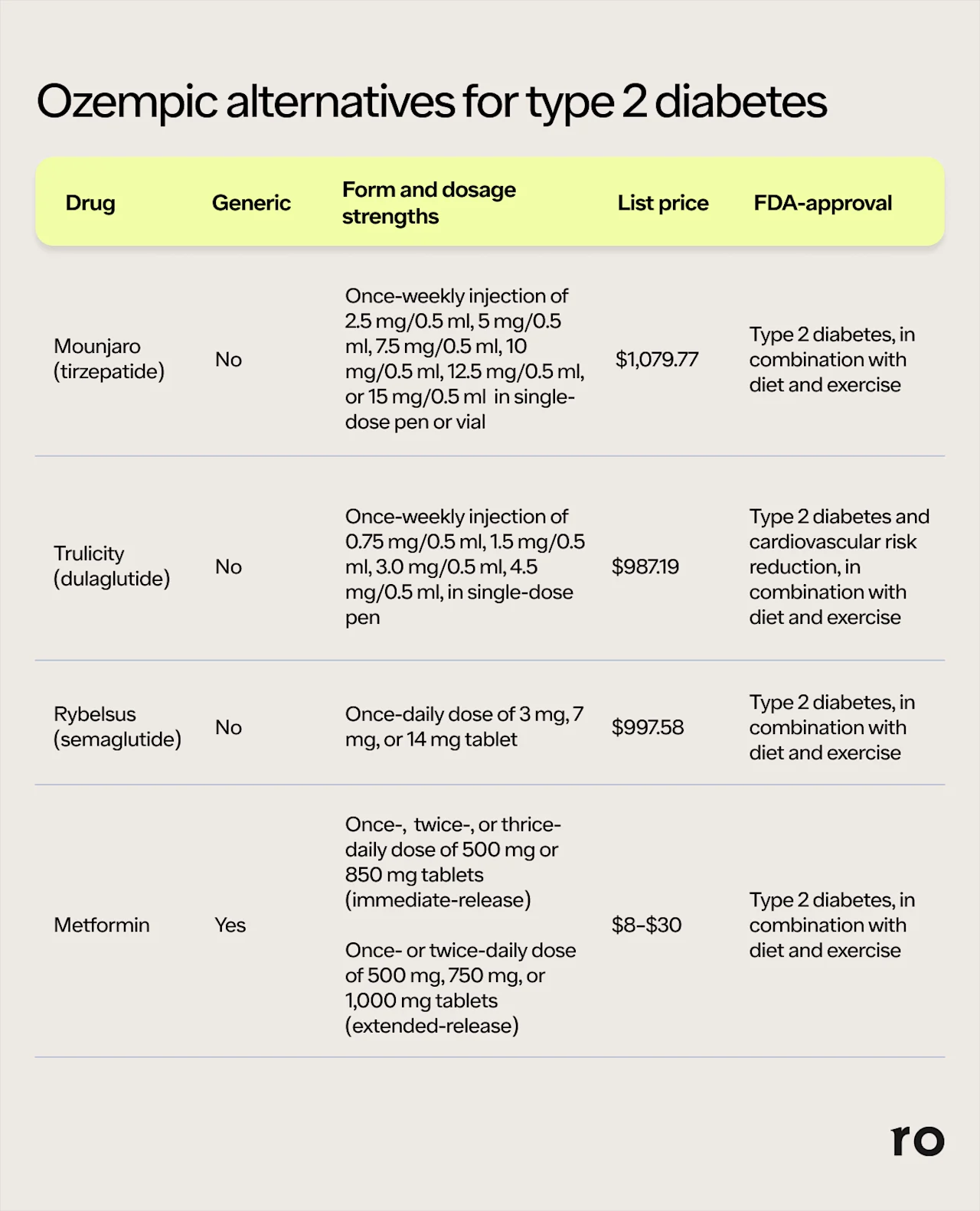
1. Mounjaro (tirzepatide)
While Mounjaro (tirzepatide) is part of the more prominent GLP-1 drug family, it differs slightly from other GLP-1 agonists like Ozempic. That’s because Mounjaro is a dual GLP-1/GIP (glucose-dependent insulinotropic polypeptide) receptor agonist, which works on two different receptors. This may make it more effective at controlling blood sugar levels and subsequent weight loss.
Like Ozempic, Mounjaro is injected on a weekly basis. Unlike Ozempic, however, it’s available in six dosage strengths: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg. While Mounjaro itself is currently only FDA-approved to help control blood sugar levels, the active ingredient in the medication, tirzepatide, has been approved for weight loss under a different name. (More on that in the next section.)
Cost: $1,079.77 for a month’s supply without insurance
2. Trulicity (dulaglutide)
Trulicity (dulaglutide) is another injectable medication that helps improve blood sugar levels and weight loss. It should be injected weekly and prescribed in addition to lifestyle changes like diet and exercise.
Like Ozempic, Trulicity is often prescribed to people with type 2 diabetes who have not found other treatments successful. The drug works by encouraging your pancreas to make more insulin when your body senses that you’ve eaten glucose or carbohydrates. Trulicity also reduces the risk that people with type 2 diabetes — both with and without heart disease — will experience a major cardiac event such as stroke, heart attack, or death.
Cost: $987.19 per month without insurance
3. Rybelsus (semaglutide)
Rybelsus has the same active ingredient as Ozempic: semaglutide. You usually take Rybelsus once a day in the morning and at least 30 minutes before you eat or drink anything. Rybelsus and Ozempic are both brand names of the same drug, but the biggest difference is that you take Rybelsus, which is available in 3 mg, 7 mg, and 14 mg tablets, by mouth, whereas Ozempic is an injection.
Cost: $997.58 per month without insurance
4. Metformin
Metformin, also known by its brand name, Glucophage, is FDA-approved as a diabetes medication and shares some side effects with Ozempic. But that’s where their similarities stop.
Metformin is available as a tablet, liquid, or in an extended-release (ER) dosage form, and depending on the formulation, the dosage strengths and how often you take the medication differ.
For example, the immediate-release tablets and liquids are often taken twice a day and come in 500 mg, 850 mg, and 1,000 mg. The ER version — which is available in 500 mg, 750 mg, and 1,000 mg — tends to be just once a day.
Metformin increases your body’s own production of GLP-1, potentially mimicking some of the effects of GLP-1 receptor agonists. The drug lowers the amount of glucose in your blood by blocking glucose production by the liver.
Metformin might also decrease blood sugar by enhancing muscle tissue’s ability to remove glucose from the bloodstream and use it for energy. Additionally, it stimulates bacteria in your gut to use more glucose, thus decreasing blood sugar levels.
And, last but certainly not least, the medication increases your body’s insulin sensitivity (i.e. your body’s ability to use insulin).
Cost: anywhere from $8 to $30 for 60 tablets with a 500 mg dosage without insurance, according to GoodRx.
Ozempic alternatives for weight loss
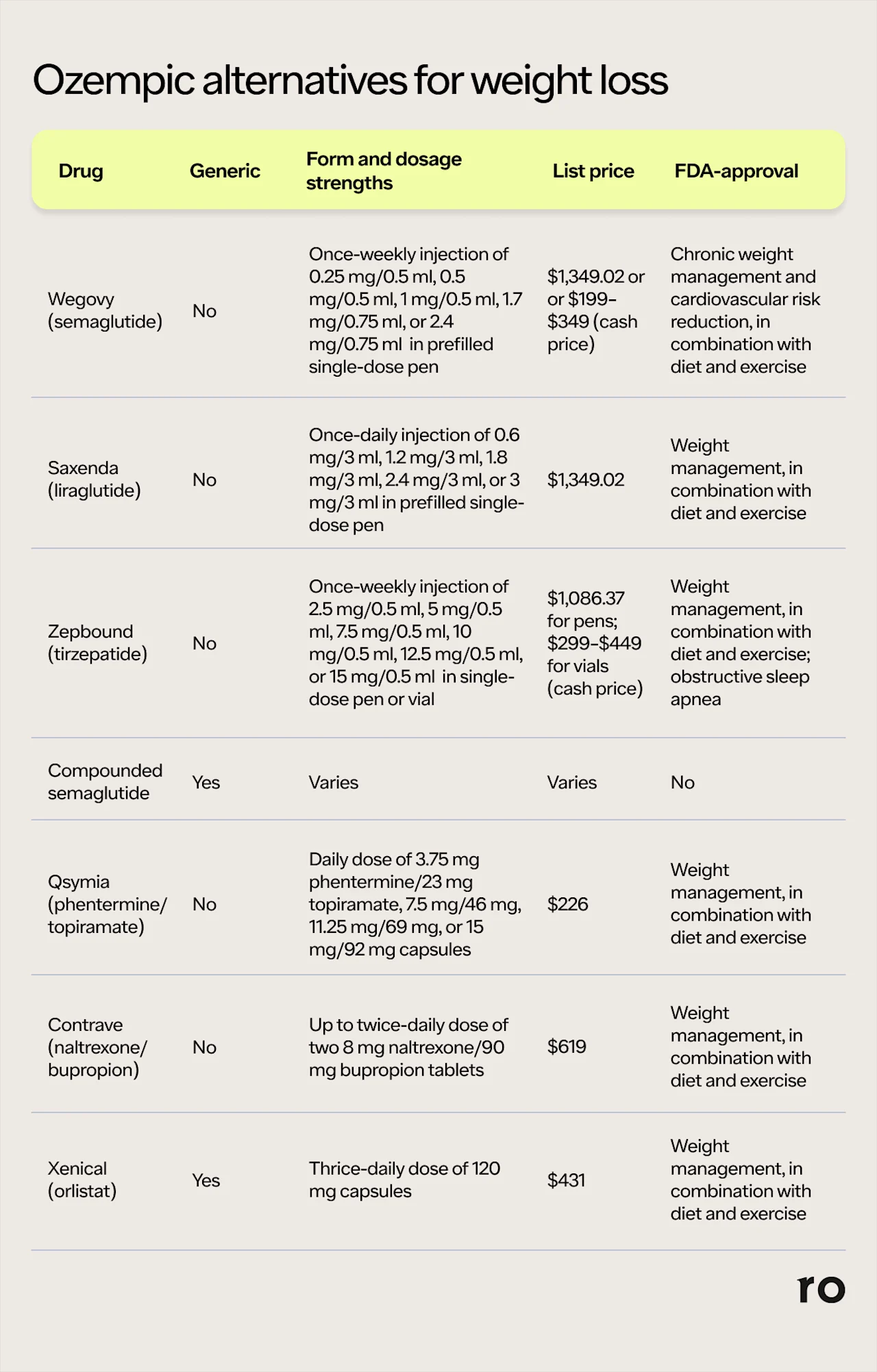
5. Wegovy (semaglutide)
Being that it also contains semaglutide, the way in which Wegovy works is the same as Ozempic. By ramping up the body’s levels of GLP-1, Wegovy keeps food in the stomach for longer and sends signals to the brain, letting it know you’re full. All of that helps regulate your appetite and leads to significant weight loss.
Wegovy is FDA-approved for weight loss and management in adults and children ages 12+ with obesity as well as adults with obesity or overweight who have at least one weight-related health condition (e.g. high blood pressure, diabetes). It can also be prescribed to lower the risk of major cardiovascular problems (e.g. heart attack, stroke) in adults with heart disease and obesity or overweight, even without type 2 diabetes.
In a clinical trial, participants who took Wegovy combined with lifestyle changes lost an average of 15% of their body weight by the end of the 68-week trial.
Cost: $1,349.02 per month without insurance or $199–$349 per month if paying with cash. (Head’s up: The $199 per month offer is only available for a limited time — until March 31, 2026 — and is only valid for the 0.25 mg and 0.5 mg doses of the drug.)
6. Saxenda (liraglutide)
Saxenda (liraglutide) is FDA-approved for chronic weight management. It also improves blood sugar control, although it is not FDA-approved to treat type 2 diabetes.
Unlike Ozempic, Saxenda is a daily injection for weight loss whose dosage strengths range from 0.6 mg to 3 mg. Weight loss on Saxenda tends to be less profound compared to that on other Ozempic alternatives that are approved for weight loss, such as Wegovy and Zepbound.
But the effects are still impressive. In a clinical trial, the majority of participants lost 5%–10% of their body weight while on Saxenda over the course of one year, and some people even lost more than 10%.
Cost: $1,349.02 per month without insurance
7. Zepbound (tirzepatide)
The latest weight loss drug to join the party, so to speak, is Zepbound: the brand name of tirzepatide. It has the same active ingredient as Mounjaro, so the two medications share many similarities. Both are GIP/GLP-1 receptor agonists, injected on a weekly basis, available in the same range of dosage strengths, and have similar side effects.
What is the key difference between the two Ozempic alternatives? Mounjaro is FDA-approved to treat type 2 diabetes, while Zepbound is FDA-approved for chronic weight management and obstructive sleep apnea.
The higher the dose of Zepbound, the more weight you’re likely to lose, according to research. In clinical trials, for example, patients taking 5 mg of Zepbound lost 15% of their body weight, patients taking 10 mg of Zepbound lost 19.5% of their body weight, and patients taking 15 mg of Zepbound lost 20.9% of their body weight after a 72-week period.
Cost: $1,086.37 per month without insurance for pens or $299–$449 per month without insurance for vials (cash price)
8. Compounded semaglutide
Compounded medications are custom-made pharmaceutical products by licensed pharmacists to meet the unique needs of an individual patient when a commercially available drug cannot. Although compounded drugs are permitted to be prescribed under federal law, they are not FDA-approved and do not undergo safety, effectiveness, or manufacturing review.
Compounded semaglutide is a weekly injection that requires a prescription from a healthcare provider. If you and your practitioner decide that compounded semaglutide is the best Ozempic alternative for you, it’s important to ensure that the medication is prepared by a reputable compounding pharmacy that adheres to strict quality control measures.
Cost: varies
9. Non GLP-1s (Qsymia, Contrave, Xenical)
While they may be skyrocketing in popularity, GLP-1s aren’t the only option for weight loss medications. Other drugs that are also FDA-approved for weight loss include:
All of these are oral medications that you take once a day.
Similar to GLP-1s, these Ozempic alternatives may cause gastrointestinal distress as well as other digestive side effects. And depending on the specific medication, people might also experience headaches, insomnia, dizziness, tingling sensations, anxiety, and irritability.
In clinical trials, the three weight loss pills have been shown to be effective. In one year, people taking Qsymia lost an average of 19 pounds, Contrave lost an average of 11 pounds, and Xenical lost an average of almost 6 pounds.
Cost:
Qsymia: $226 for 30 capsules without insurance
Contrave: $619 for 120 tablets without insurance
Xenical: $431 for 90 capsules without insurance
What Ozempic alternative is right for me?
Short answer: It depends.
If Ozempic isn’t available to you, either because of a shortage or because your insurance doesn’t cover it, your healthcare provider will help you find the right Ozempic alternative, factoring in your age, health situation, treatment goals, and personal preferences.
How to switch from Ozempic to an alternative medication?
If you’re interested in switching from Ozempic to an alternative medication, make an appointment with a healthcare provider. They can determine which Ozempic alternative (if any) could be a good option for you and offer guidance on how to switch from one drug to the other.
Your healthcare provider may recommend slowly tapering off Ozempic before starting you on the new medication in order to minimize side effects. Follow their medical advice to ease the transition from Ozempic to the alternative.
Rx weight loss with Ro
Get access to prescription weight loss medication online
Bottom line
If Ozempic isn’t available to you — whether due to cost, coverage, or supply issues — there are other medications that may help with blood sugar control and weight loss. Alternative treatments include injectable GLP-1 medications as well as oral tablets and capsules. Talking with a healthcare provider is key to figuring out which option fits you and your goals. But here are a few takeaways to keep in mind:
Ozempic is just one of many GLP-1s approved for type 2 diabetes. Other options include Mounjaro, Trulicity, and Rybelsus.
Some alternatives are FDA-approved for weight loss and management. Wegovy and Zepbound are weekly injections, while Saxenda is daily.
Oral medications may be a fit for diabetes or weight loss. Pills like metformin, Qsymia, Contrave, and Xenical offer non-injectable alternatives with different mechanisms of action.
Cost and insurance coverage of Ozempic alternatives can vary widely, with exact prices depending on factors like location, pharmacy, insurance plan, and, of course, the specific drug. Out-of-pocket costs can range from as little as $8 to over $1,300 per month.
Lifestyle still matters. No matter the medication you — alongside your healthcare provider — decide to try, it should still be combined with a nutritious diet and regular exercise.
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
Ozempic Important Safety Information: Read more about serious warnings and safety info.
GLP-1 Important Safety Information: Read more about serious warnings and safety info.
Mounjaro Important Safety Information: Read more about serious warnings and safety info.
Wegovy Important Safety Information: Read more about serious warnings and safety info.
Saxenda Important Safety Information: Read more about serious warnings and safety info.
Zepbound Important Safety Information: Read more about serious warnings and safety info.
References
American Pharmacists Association. (2021). Frequently Asked Questions About Pharmaceutical Compounding. Retrieved from https://www.pharmacist.com/Practice/Patient-Care-Services/Compounding/Compounding-FAQs
Contrave. (2024). CurAccess Program: Pay $99 Or Less + FREE Shipping.* Retrieved from https://contrave.com/save/
Corcoran, C. & Jacobs, T. F. (2023). Metformin. StatPearls. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK518983/
Drugs.com-a. (n.d.). Comparing Alli vs Xenical. Retrieved from https://www.drugs.com/compare/alli-vs-xenical
Drugs.com-b. (n. d.). Contrave Prices, Coupons, Copay Cards, and Patient Assistance Programs. Retrieved from https://www.drugs.com/price-guide/contrave
Drugs.com-c. (n.d.). Qsymia Prices, Coupons, Copay Cards, and Patient Assistance Programs. Retrieved from https://www.drugs.com/price-guide/qsymia
Drugs.com-d. (n. d.). Xenical Prices, Coupons, Copay Cards, and Patient Assistance Programs. Retrieved https://www.drugs.com/price-guide/xenical
Eli Lilly-a. (n.d.). LillyDirect. Retrieved from https://lillydirect.lilly.com/
Eli Lilly-b. (n. d.). Mounjaro Pricing Information. Retrieved from https://pricinginfo.lilly.com/mounjaro
Eli Lilly-c. (n. d.). Trulicity Pricing Information. Retrieved from https://pricinginfo.lilly.com/trulicity
GoodRx. (2024). Metformin Prices, Coupons & Savings Tips. Retrieved from https://www.goodrx.com/metformin
Idrees, Z., Cancarevic, I., & Huang, L. (2022). FDA-Approved Pharmacotherapy for Weight Loss Over the Last Decade. Cureus, 14(9), e29262. doi: 10.7759/cureus.29262. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9579826/
Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. The New England Journal of Medicine, 387(3), 205–216. doi: 10.1056/NEJMoa2206038. Retrieved from https://www.nejm.org/doi/10.1056/NEJMoa2206038
Khera, R., Murad, M. H., Chandar, A. K., et al. (2016). Association of Pharmacological Treatments for Obesity With Weight Loss and Adverse Events: A Systematic Review and Meta-analysis. JAMA, 315(22), 2424–2434. doi: 10.1001/jama.2016.7602. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5617638/
Mehta, A., Marso, S. P., & Neeland, I. J. (2017). Liraglutide for weight management: a critical review of the evidence. Obesity Science & Practice, 3(1), 3–14. doi: 10.1002/osp4.84. Retrieved from https://pubmed.ncbi.nlm.nih.gov/28392927/
Nauck, M. A. & D'Alessio, D. A. (2022). Tirzepatide, a dual GIP/GLP-1 receptor co-agonist for the treatment of type 2 diabetes with unmatched effectiveness regrading glycaemic control and body weight reduction. Cardiovascular Diabetology, 21(1), 169. https://doi.org/10.1186/s12933-022-01604-7. Retrieved from https://cardiab.biomedcentral.com/articles/10.1186/s12933-022-01604-7
NovoCare-a. (2023). Saxenda (liraglutide) injection 3 mg list price & Insurance Coverage explained: NovoCare. Retrieved from https://www.novocare.com/obesity/products/saxenda/explaining-list-price.html
NovoCare-b. (2023). Wegovy (semaglutide) injection 2.4 mg list price & Insurance Coverage explained: NovoCare. Retrieved from https://www.novocare.com/obesity/products/wegovy/let-us-help/explaining-list-price.html
NovoCare. (2024). What is the list price for Rybelsus and will it impact me?. Retrieved from https://www.novocare.com/diabetes/products/rybelsus/explaining-list-price.html
O'Neil, P. M., Birkenfeld, A. L., McGowan, B., et al. (2018). Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: a randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. Lancet (London, England), 392(10148), 637–649. doi: 10.1016/S0140-6736(18)31773-2. Retrieved from https://pubmed.ncbi.nlm.nih.gov/30122305/
Preiss, D., Dawed, A., Welsh, P., et al. (2017). Sustained influence of metformin therapy on circulating glucagon-like peptide-1 levels in individuals with and without type 2 diabetes. Diabetes, Obesity & Metabolism, 19(3), 356–363. doi: 10.1111/dom.12826. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5330429/
U.S. Food and Drug Administration (FDA). (n.d.). Alli Orlistat Label. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/021887lbl.pdf
U.S. Food and Drug Administration (FDA-a). (2025). Highlights of Prescribing Information: Contrave (naltrexone- hydrochloride and bupropion hydrochloride) extended-release tablets, for oral use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/200063s024s026lbl.pdf
U.S. Food and Drug Administration (FDA). (2018). Highlights of Prescribing Information: Glucophage (metformin hydrochloride) tablets, for oral use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/020357s034,021202s018lbl.pdf
U.S. Food & Drug Administration (FDA-b). (2025). Highlights of Prescribing Information: Mounjaro (tirzepatide) Injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/217806s031lbl.pdf
U.S. Food and Drug Administration (FDA-c). (2025). Highlights of Prescribing Information: Rybelsus (semaglutide) tablets, for oral use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/213051s024,s028s029lbl.pdf
U.S. Food and Drug Administration (FDA-d). (2025). Highlights of Prescribing Information: Saxenda (liraglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/206321s022lbl.pdf
U.S. Food and Drug Administration (FDA-e). (2025). Highlights of Prescribing Information: Trulicity (dulaglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/125469s063lbl.pdf
U.S. Food and Drug Administration (FDA). (2024). Highlights of Prescribing Information: Qsymia (phentermine and topiramate extended-release capsules), for oral use, CIV. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/022580s026lbl.pdf
U.S. Food & Drug Administration (FDA-f). (2025). Highlights of Prescribing Information: Wegovy (semaglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215256s015lbl.pdf
U.S. Food and Drug Administration (FDA-g). (2022). Highlights of Prescribing Information: Xenical (orlistat) Capsules for oral use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020766s038lbl.pdf
U.S. Food & Drug Administration (FD-hA). (2025). Highlights of Prescribing Information: Zepbound (tirzepatide) Injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/217806s031lbl.pdf
Wilding, J., Batterham, R. L., Calanna, S., et al. (2021). Once-weekly semaglutide in adults with overweight or obesity. The New England Journal of Medicine, 384(11), 989–1002. doi: 10.1056/NEJMoa2032183. Retrieved from https://pubmed.ncbi.nlm.nih.gov/33567185/