Key takeaways
Ozempic overdose is possible and can lead to severe nausea, vomiting, abdominal pain, and, in rare cases, pancreatitis or intestinal blockage.
If you take too much Ozempic, call the Poison Help line at 1-800-222-1222.
Here's what we'll cover
Key takeaways
Ozempic overdose is possible and can lead to severe nausea, vomiting, abdominal pain, and, in rare cases, pancreatitis or intestinal blockage.
If you take too much Ozempic, call the Poison Help line at 1-800-222-1222.
While Ozempic (semaglutide) is typically self-administered using a prefilled injection pen, it is possible to accidentally give yourself a higher dose than prescribed. So what happens if you take too much Ozempic? Is it possible to experience an Ozempic overdose?
Read on as we explain what to do if you take too much Ozempic, what an Ozempic overdose can look like, and when to seek emergency medical attention.
Can you overdose on Ozempic?
Yes, it is possible to overdose on Ozempic. Meaning you can take a higher dose of Ozempic than you’ve been prescribed, and doing so can lead to side effects ranging in severity.
An Ozempic overdose can cause significant nausea, diarrhea, vomiting, and gastrointestinal symptoms, says Supriya Rao, MD, DABOM, a quadruple board-certified physician specializing in obesity medicine and lifestyle medicine. “Usually, if you accidentally take two doses, you’ll feel a little sicker than normal, but from what I’ve seen with patients, they’re able to manage symptoms at home.”
That being said, pancreatitis is also possible. This “is an inflammation with severe abdominal pain that requires hospital admission where you can be given IV fluids,” Dr. Rao explains. “Rarely, you can get an intestinal blockage, or ileus, because the medication works by slowing down your digestion. Ileus is not a physical blockage but a functional one where the colon doesn’t work the way it should and acts as if there’s a blockage. There are also rare reports of seizures.”
Since 2019, America’s Poison Centers have seen a nearly 1,500% increase in calls related to overdose or side effects of injectable GLP-1 medications like Ozempic. Some of that increase may be explained by the increase in usage of these drugs: Between 2019 and 2020 alone, Ozempic usage increased by over 1,200%, according to one analysis by the American Pharmacists Association. Between 2020 and 2022, the number of people taking Ozempic nearly tripled.

What happens if you take too much Ozempic?
According to the FDA and the American Pharmacists Association, symptoms of Ozempic overdose may include:
Severe vomiting
Severe hypoglycemia (low blood sugar levels)
Abdominal pain
Fainting
Headache
Migraine
Dehydration
Weakness
Loss of appetite
Acute pancreatitis
Gallstones
Some of these gastrointestinal symptoms, such as nausea and vomiting, can last for days, and some cases of Ozempic overdose require hospitalization. There, you might be given anti-nausea medication and/or intravenous (IV) fluids. Because semaglutide’s effects can last about one week, some people may need to stay in the hospital for treatment and observation for a while.
As Dr. Rao shares above, taking an extra dose of Ozempic may make you feel “sicker than normal,” but many patients are able to manage symptoms at home. “If they’re experiencing significant GI symptoms, I’ll send them to the ER to get fluids and be treated for those symptoms more acutely,” she adds.
What should you do if you take too much Ozempic or experience Ozempic overdose?
If you take too much Ozempic, contact the Poison Help line at 1-800-222-1222. They can advise you on next steps based on your symptoms. You can also give your healthcare provider a call and “tell them your symptoms, especially if they’re getting progressively worse or not improving at all,” Dr. Rao says.
“It’s one thing if you’re feeling sick or not so great,” she continues. “It’s another if you’re feeling extreme pain or like you’re going to pass out, you can’t stop vomiting or have severe abdominal pain, or you can’t pass stool or haven’t had a bowel movement in a few days.” Those are serious symptoms that warrant emergency medical attention, Dr. Rao explains.
Recommended Ozempic dosages
The recommended Ozempic dosage depends on why you’re taking it. While commonly used off-label for weight loss, the FDA has approved Ozempic for the following uses, along with diet and exercise:
To control blood sugar levels in adults with type 2 diabetes
To lower the risk of serious health problems, such as heart attack, in adults with type 2 diabetes and heart disease
To lower the risk of worsening disease, complications. and death in adults with type 2 diabetes and chronic kidney disease
If you’re prescribed Ozempic, you will give yourself a weekly subcutaneous injection (under the skin of the upper arm, thigh, or stomach). Ozempic is available in prefilled pens. The Ozempic pens contain multiple doses of the medication, and you’ll select your dose using the dial on the pen.
Whether you’re taking Ozempic off-label for weight loss or to manage type 2 diabetes, kidney disease, or heart disease, your healthcare provider will likely start you on the lowest dose of 0.25 mg to get your body used to the medication. Then, over a period of weeks, they’ll slowly increase the dose until you reach a maintenance dosage that is effective for you while keeping side effects manageable. The maximum dose of Ozempic for type 2 diabetes is 2 mg; for those who also have heart or kidney disease, it’s 1 mg.
While everyone is different and dosing will be customized to your needs (e.g. additional blood sugar control), the typical Ozempic dosing schedule may look like this:
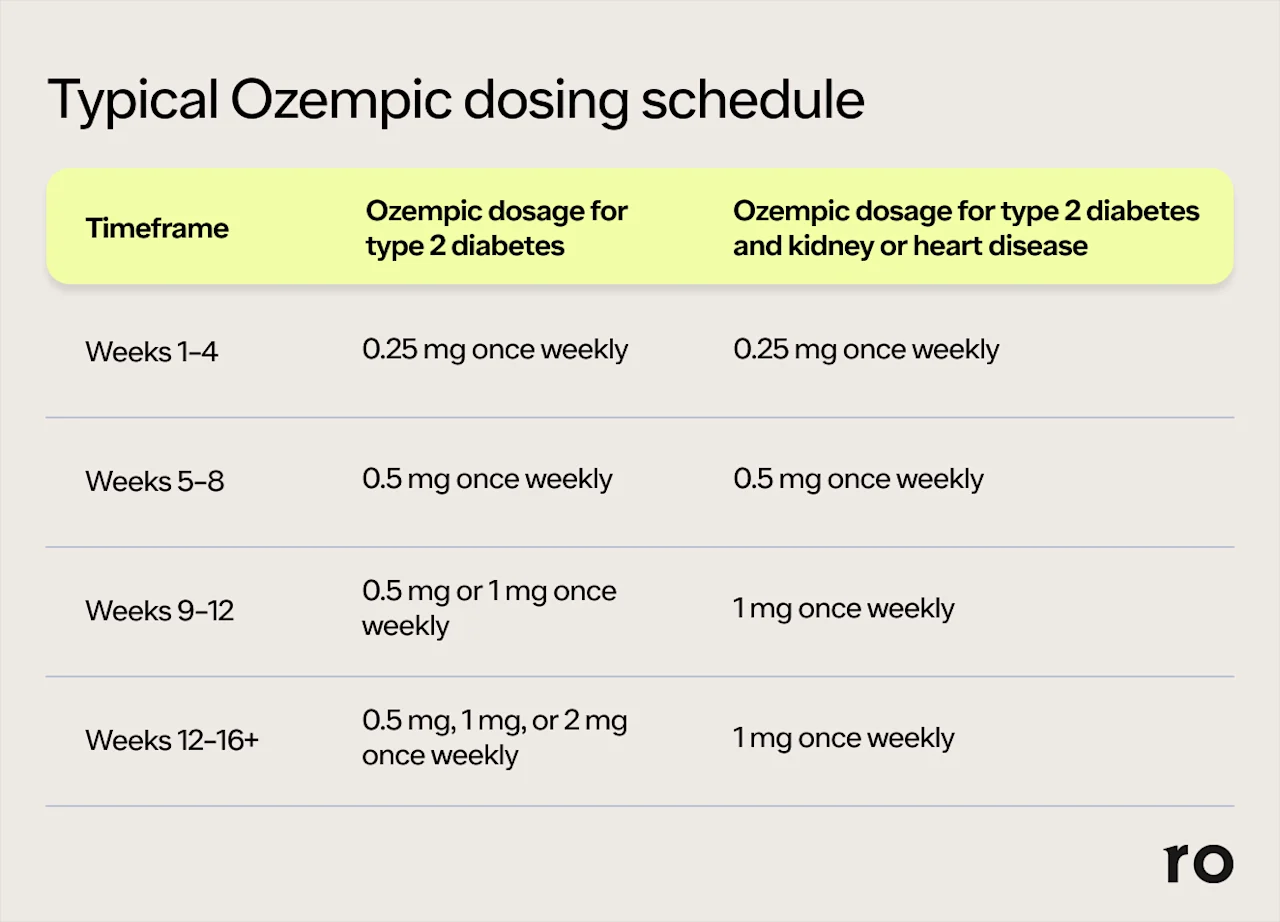
Remember: Your prescribed dose of Ozempic is the one that your healthcare provider has determined to be most effective and the safest for you. Taking more Ozempic will not make it work more effectively. It will, however, make you more likely to experience side effects, some of which could become potentially serious. Before you reach your maintenance dose, it’s important to be patient and follow the dosing schedule prescribed by your doctor. This is designed to help your body adjust to the medication and better cope with the side effects of Ozempic.
How to avoid taking too much Ozempic
To avoid taking too much Ozempic, you can make a few small tweaks to your dosing routine. These shouldn’t take too much time, and they can help ensure you inject Ozempic safely.
When using an Ozempic pen, check that your pen shows the correct dose on the label. Then, select your dose by turning the dose selector until your dose appears in the dose counter. It should line up exactly with the tip of the triangle next to the letters “mg.”
“The safety and efficacy of these medications is as good as whoever is prescribing them and seeing you, and how good you are at adhering to the recommended lifestyle changes,” Dr. Rao says.“Being a good advocate for yourself and having an open dialogue with your physician is also important.”
Rx weight loss with Ro
Get access to prescription weight loss medication online
Can you overdose on other semaglutide drugs?
Yes, it is possible to overdose on other semaglutide drugs besides Ozempic, including Wegovy (a weekly injection) and Rybelsus (a daily oral tablet). Symptoms of semaglutide overdose may include severe nausea, vomiting, and hypoglycemia, and some cases may require hospitalization. If you take too much of Wegovy or Rybelsus, contact the Poison Help line at 1-800-222-1222 as well as your healthcare provider.
Bottom line
While accidental overdoses are relatively rare with the prefilled Ozempic pens, errors can occur. To stay safe, it’s essential to follow your prescribed dosage, know the signs of a possible Ozempic overdose, and seek medical help when necessary. Here’s a quick recap of what happens if you take too much Ozempic and other key points we covered above:
Taking more than your prescribed dose can lead to severe gastrointestinal distress or hypoglycemia, and, in rare cases, complications like pancreatitis or ileus.
Ozempic overdose symptoms, including severe nausea, vomiting, and abdominal pain, may persist for around a week due to the drug’s long half-life.
Serious symptoms, such as extreme abdominal pain, persistent vomiting, or an inability to pass stool, may require hospitalization and should lead you to seek emergency medical attention.
Always double-check your dose before injecting, and consult your healthcare provider if you’re unsure about your dosing instructions. If you take too much Ozempic, call the Poison Help line at 1-800-222-1222.
Frequently asked questions (FAQs)
What happens if you inject Ozempic twice?
If you inject Ozempic twice, you may experience more intense side effects than usual, including nausea, vomiting, and abdominal pain. If symptoms do not dissipate or worsen, seek emergency medical attention. If you accidentally take two doses of Ozempic, contact the Poison Help line at 1-800-222-1222. It’s also a good idea to contact your healthcare provider (as is always the case when it comes to questions or concerns about a prescribed medication).
What is the highest amount of Ozempic you can take?
The maximum dose of Ozempic for type 2 diabetes is 2 mg weekly. However, your healthcare provider may prescribe a lower dose of 1 mg or 0.5 mg if that effectively controls your blood sugar levels. The maximum dose of Ozempic for kidney issues or heart disease is 1 mg weekly. Never take more of the drug than prescribed.
How do you flush Ozempic out of your system?
Ozempic has a half-life of around one week. That means that every seven days, about half of the dose will flush out of your system. So, it can take up to five weeks (or 35 days) for the medication to fully leave your body after you stop taking Ozempic.
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
Ozempic Important Safety Information: Read more about serious warnings and safety info.
GLP-1 Important Safety Information: Read more about serious warnings and safety info.
Wegovy Important Safety Information: Read more about serious warnings and safety info.
References
2023 ACMT Annual Scientific Meeting Abstracts – San Diego, CA. (2023). Journal of Medical Toxicology, 19(2), 63–168. doi: 10.1007/s13181-023-00930-w. Retrieved from https://link.springer.com/article/10.1007/s13181-023-00930-w
America’s Poison Centers. (2025). Glucagon-like Peptide-1 (GLP-1) Agonists. National Poison Data System. Retrieved from https://poisoncenters.org/track/GLP-1
Kommu, S. & Whitfield, P. (2024). Semaglutide. StatPearls. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK603723/
Lambson, J. E., Flegal, S. C., & Johnson, A. R. (2023). Administration errors of compounded semaglutide reported to a poison control center-Case series. Journal of the American Pharmacists Association: JAPhA, 63(5), 1643–1645. doi: 10.1016/j.japh.2023.06.017. Retrieved from https://pubmed.ncbi.nlm.nih.gov/37392810/
Novo Nordisk. (2024). Dosing Schedule | Ozempic (semaglutide) injection. Retrieved from https://ro.co/weight-loss/ozempic-side-effects/
Smits, M. M. & Van Raalte, D. H. (2021). Safety of Semaglutide. Frontiers in Endocrinology, 12, 645563. doi: 10.3389/fendo.2021.645563. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC8294388/
U.S. Food and Drug Administration (FDA-a). (2024). FDA alerts health care providers, compounders and patients of dosing errors associated with compounded injectable semaglutide products. Retrieved from https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-patients-dosing-errors-associated-compounded
U.S. Food and Drug Administration (FDA). (2025). Highlights of Prescribing Information: Ozempic (semaglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl.pdf
U.S. Food and Drug Administration (FDA-b). (2024). Highlights of Prescribing Information: Rybelsus (semaglutide) tablets, for oral use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/213051Orig1s020,213051Orig1s021lbl.pdf
U.S. Food and Drug Administration (FDA-c). (2024). Highlights of Prescribing Information: Wegovy (semaglutide) injection, for subcutaneous use. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215256s015lbl.pdf
Watanabe, J. H., Kwon, J., Nan, B., et al. (2024). Trends in glucagon-like peptide 1 receptor agonist use, 2014 to 2022. Journal of the American Pharmacists Association: JAPhA, 64(1), 133–138. doi: 10.1016/j.japh.2023.10.002. Retrieved from https://pubmed.ncbi.nlm.nih.gov/37821008/